
Concept explainers
Interpretation:
Identification of the given liquid is to be done and the chemical equation has to be written.
Concept introduction:
The given liquid can be either benzene or cyclohexene. The reaction of bromine vapour with an alkene gives decolorisation of bromine vapours. This reaction is used for the test of unsaturation or double bond in hydrocarbons.
Bromination of
Alkene undergoes bromination which yields the dibromo compound (vicinal dibromides or 1,2-dibromides).
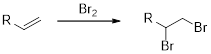
Benzene does not react with bromine vapours. However, it undergoes electrophilic substitution reaction with halogens in presence of a lewis acid such as
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Alcohols are very useful starting materials for the production of many different compounds. The following conversions, starting with 1-butanol, can be carried out in two or more steps. Show the steps (reactants/catalysts) you would follow to carry out the conversions, drawing the formula for the organic product in each step. For each step, a major product must be produced. (See Exercise 62.) (Hint: In the presence of H+, an alcohol is converted into an alkene and water. This is the exact reverse of the reaction of adding water to an alkene to form an alcohol.) a. 1-butanol butane b. 1-butanol 2-butanonearrow_forwardComplete and balance the following combustion reactions. Assume that each hydrocarbon is converted completely to carbon dioxide and water. (a) Propane + O2 (b) Octane + O2 (c) Cyclohexane + O2 (d) 2-Methylpentane + O2arrow_forwardDefine the Properties of Alkyl Halides ?arrow_forward
- What is the balanced chemical equation when gaseous butanol, C4H9OH, undergoes combustion?arrow_forwardName the major chemical groups found in organic molecules. Describe the basic structure of each chemical group and outline the chemical properties of the organic molecules in which they occur.arrow_forwardWhat characteristics of carbon make it ideal for the formation of organic compounds?arrow_forward
- Identify the characteristics of phenolic compounds and alcohols,explain how they act, and evaluate their primary uses.arrow_forwardSalicylic acid is added into a test tube, followed by methanol and sulfuric acid as the catalyst. It is heated for 10-15 minutes and poured into a beaker with crushed ice. This is the esterification of Oil of wintergreen. Write the complete reaction equation and describe the odor of the reactants (salicylic acid and methanol) and the products (oil of wintergreen).arrow_forwardWhat are the solubility properties of organic compounds?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





