
Concept explainers
(a)
Interpretation:
The hybrid orbitals on the central atom that form bond in
Concept Introduction:
Hybridization is the idea that atomic orbitals combine to form new hybridized orbitals which in turn, influences molecular geometry and bonding properties. Hybridization is also an expansion of the
(a)
Answer to Problem 10.57QE
The hybrid orbitals on the central atom that form bond in
Explanation of Solution
The structure of
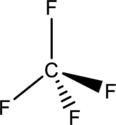
The
The process of mixing and rearrangement of one s and three p orbitals of valence shell of same atom to form new four hybrid orbitals having maximum symmetry and definite orientation is known as
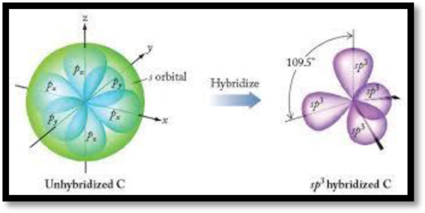
Figure 1
(b)
Interpretation:
The hybrid orbitals on the central atom that form bond in
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 10.57QE
The hybrid orbitals on the central atom that form bond in
Explanation of Solution
The structure of
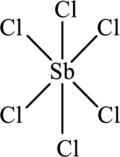
The
The
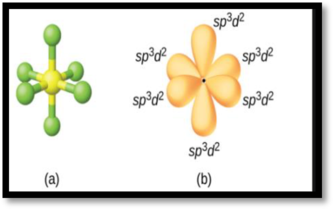
Figure 2
(c)
Interpretation:
The hybrid orbitals on the central atom that form bond in
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 10.57QE
The hybrid orbitals on the central atom that form bond in
Explanation of Solution
The structure of
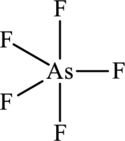
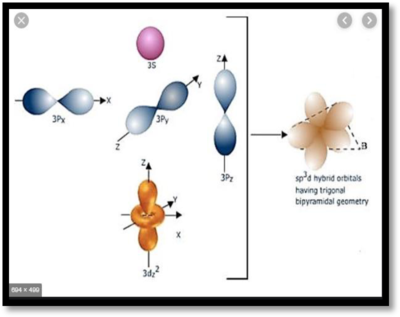
Figure 3
(d)
Interpretation:
The hybrid orbitals on the central atom that form bond in
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 10.57QE
The hybrid orbitals on the central atom that form bond in
Explanation of Solution
The structure of
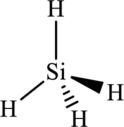
The
The process of mixing and rearrangement of one s and three p orbitals of valence shell of same atom to form new four hybrid orbitals having maximum symmetry and definite orientation is known as
(e)
Interpretation:
The hybrid orbitals on the central atom that form bond in
Concept Introduction:
Refer to part (a).
(e)
Answer to Problem 10.57QE
The hybrid orbitals on the central atom that form bond in
Explanation of Solution
The structure of
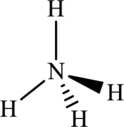
The
The process of mixing and rearrangement of one s and three p orbitals of valence shell of same atom to form new four hybrid orbitals having maximum symmetry and definite orientation is known as
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: Principles and Practice
- 4. (a) Draw the shape of the atomic valence orbitals formed by the overlaping of two fluoride 2p atomic orbitals. (b) Draw the molecular orbital diagrams for F2 and F2*. Identify their bond order and magnetic properties. (c) An unstable nucleus exhibit radioactivity. (i) Explain how the number of protons and neutrons in a radioactive nucleus can be used to predict its probable mode decay. (ii) Illustrate your answer in (i) with a schematic graph.arrow_forwardPredict the electron pair geometry and the molecular structure of each of the following molecules or ions:(a) SF6(b) PCl5(c) BeH2(d) CH3+arrow_forwardWhat are the electron-pair geometry and the molecular structure of each of the following molecules or ions?(a) ClF5(b) ClO2−(c) TeCl42−(d) PCl3(e) SeF4(f) PH2−arrow_forward
- . Assume that the third-period element phosphorus forms a diatomic molecule, P2, in an analogous way as nitrogen does to form N2. (a) Write the electronic configuration for P2. Use [Ne2] to represent the electron configuration for the first two periods. (b) Calculate its bond order. (c) What are its magnetic properties (diamagnetic or paramagnetic)?arrow_forwardChemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is isoelectronic with them. (a) CN–, (b) NH4+ (c) CO3 2–arrow_forwardWhat is the hybridization of the central atom in each of the following?(a) BeH2(b) SF6(c) PO4 3−(d) PCl5arrow_forward
- (a) Methane (CH4) and the perchlorate ion (ClO4- ) are bothdescribed as tetrahedral. What does this indicate about theirbond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?arrow_forward10.What is the hybridization of the central atom in each of the following? (a) BeH2 (b) SF6 (c) PO43− (d) PCl5arrow_forwardGive the hybridization of the metalloid and the molecular geometry for each of the following compounds or ions. (a) GeH4(b) SbF3(c) Te(OH)6(d) H2Te(e) GeF2(f) TeCl4(g) SiF62−(h) SbCl5(i) TeF6arrow_forward
- The structure of caffeine is shown below. (a) Complete the Lewis structure. (b) How many pi bonds are present in caffeine? How many sigma bonds? (c) Identify the hybridization of the carbon atoms. (d) What is the value of the O-C-N angle?arrow_forwardIdentify the electron pair geometry and the molecular structure of each of the following molecules or ions:(a) IF6+(b) CF4(c) BF3(d) SiF5−(e) BeCl2arrow_forwardConsider the reaction BF3 + NH3 -> F3B-NH3 (a) Describe the changes in hybridization of the B and N atoms as a result of this reaction. (b) Describe the shapes of all the reactant molecules with their bond angles. (c) Draw the overall shape of the product molecule and identify the bond angles around B and N atoms. (d) What is the name of the bond between B and N. (e)Describe the bonding orbitals that make the B and F, B and N & N and H bonds in the product molecule.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
