
(a)
Interpretation:
Bond angle has to be predicted using VSEPR model for the given structure and also the hybrid orbitals on the central atoms has to be given. The molecule is polar or not also has to be indicated.
Concept Introduction:
Lewis structure is used for predicting the shape of molecules. From the steric number obtained in a Lewis structure, the molecular geometry can be predicted. VSEPR model can predict the shape of molecules considering their Lewis structure. Certain rules has to be followed in for the VSEPR model.
- The molecule will have a shape where there is minimal electrostatic repulsion between the valence‑shell electron pairs.
- The forces of repulsion between two lone pairs of electrons will be higher than the repulsion between lone pair and bond pair of electrons. This in turn will be higher than the bond pair‑bond pair of electrons.
The hybridized orbitals and the steric number can be related as shown below;
| Steric number | Hybridized orbital |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 |
(a)
Explanation of Solution
Resonance structure:
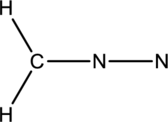
The given species is shown below;
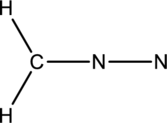
The total number of valence electrons is calculated as shown below;
A total of

Hybrid orbitals of central atoms in structure I:
The resonance structure is shown below;
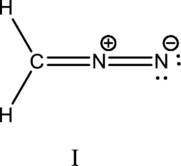
Hybrid orbitals of central nitrogen atom:
The nitrogen atom has does not have a lone pair of electrons and it is bonded to two atoms. Therefore, the steric number is calculated as shown below;
As the steric number is two, the hybridization of nitrogen atom is
Hybrid orbital of carbon atom:
The carbon atom does not have lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is three, the hybridization of carbon atom is
Hybrid orbitals of central atoms in structure II:
The resonance structure is shown below;
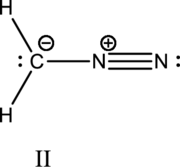
Hybrid orbitals of central nitrogen atom:
The nitrogen atom has does not have a lone pair of electrons and it is bonded to two atoms. Therefore, the steric number is calculated as shown below;
As the steric number is two, the hybridization of nitrogen atom is
Hybrid orbital of carbon atom:
The carbon atom have one lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of carbon atom is
Two resonance structures do not use the same hybrid orbitals because the hybridization of the carbon atom is different in both.
Polarity of the species:

From the above figure, it is found that there is a permanent dipole moment. Hence, the molecule will be polar.
(b)
Interpretation:
Bond angle has to be predicted using VSEPR model for the given structure and also the hybrid orbitals on the central atoms has to be given. The molecule is polar or not also has to be indicated.
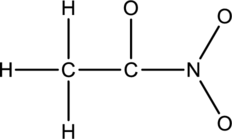
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Resonance structure:
The given species is shown below;
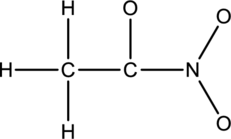
The total number of valence electrons is calculated as shown below;
A total of
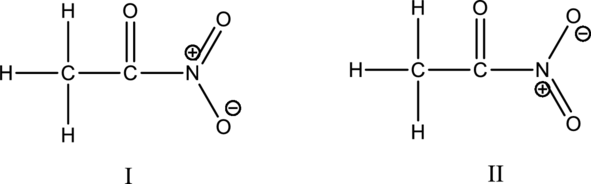
Hybrid orbitals of central atoms in structure I and II:
The resonance structures is shown below;
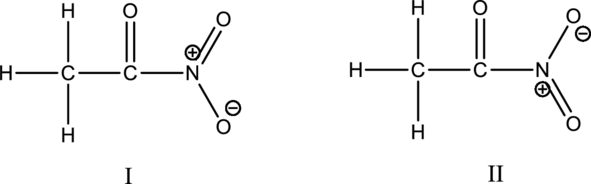
Hybrid orbitals of nitrogen atom:
The nitrogen atom has does not have a lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is three, the hybridization of nitrogen atom is
Hybrid orbital of first carbon atom:
The first carbon atom does not have lone pair of electrons and it is bonded to four atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of carbon atom is
Hybrid orbital of second carbon atom:
The second carbon atom does not have lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is three, the hybridization of carbon atom is
Two resonance structures use the same hybrid orbitals because the hybridization of the carbon atoms and nitrogen atom are same.
Polarity of the species:
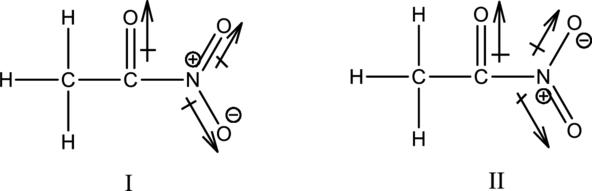
From the above figure, it is found that there is a permanent dipole moment. Hence, the molecule will be polar.
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: Principles and Practice
- Lets look more closely at the process of hybridization. (a) What is the relationship between the number of hybrid orbitals produced and the number of atomic orbitals used to create them? (b) Do hybrid atomic orbitals form between different p orbitals without involving 5 orbitals? (c) What is the relationship between the energy of hybrid atomic orbitals and the atomic orbitals from which they are formed?arrow_forwardMethylcyanoacrylate is the active ingredient in super glues. Its Lewis structure is (a) How many sigma bonds are in the molecule? (b) How many pi bonds are in the molecule? (c) What is the hybridization of the carbon atom bonded to nitrogen? (d) What is the hybridization of the carbon atom bonded to oxygen? (e) What is the hybridization of the double-bonded oxygen?arrow_forward• explain how hybridization reconciles observed molecular shapes with the orbital overlap model.arrow_forward
- 7.59 What type of hybrid orbital is generated by combining the valence s orbital and all three valence p orbitals of an atom? How many hybrid orbitals result?arrow_forward• explain the formation of multiple bonds in terms of the overlap of a combination of hybridized and unhybridized atomic orbitals.arrow_forwardIf you have three electron regions around a central atom, how can you have a triangular planar molecule? An angular molecule? What bond angles are predicted in each case?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





