
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.6, Problem 9P
Predict the products of the following polar reaction, a step in the citric acid cycle for food
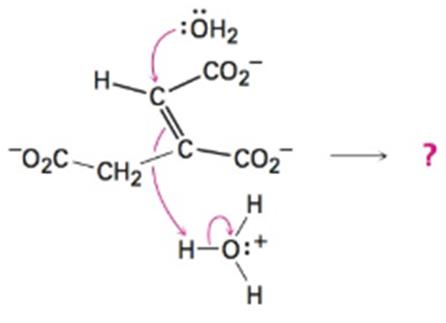
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which of the following interactions can be definitely described as electrostatic in nature?
O Hard acid - hard base
O Soft acid- soft base
O Hard acid - soft base
O Soft acid - hard abse
Please provide a drawing explaning the answer.
Write down the Structure Activity Relationship(SAR) of Tricyclic Antidepressant(TCA)
Chapter 6 Solutions
Organic Chemistry
Ch. 6.1 - Prob. 1PCh. 6.3 - Prob. 2PCh. 6.3 - Using curved fishhook arrows, propose a mechanism...Ch. 6.4 - Prob. 4PCh. 6.4 - An electrostatic potential map of boron...Ch. 6.5 - What product would you expect from reaction of...Ch. 6.5 - Reaction of HBr with 2-methylpropene yields...Ch. 6.6 - Prob. 8PCh. 6.6 - Predict the products of the following polar...Ch. 6.7 - Which reaction is more energetically favored, one...
Ch. 6.7 - Prob. 11PCh. 6.9 - Which reaction is faster, one with ∆G‡ = +45...Ch. 6.10 - Prob. 13PCh. 6.SE - Prob. 14VCCh. 6.SE - Prob. 15VCCh. 6.SE - Prob. 16VCCh. 6.SE - Look at the following energy diagram: (a) Is...Ch. 6.SE - Look at the following energy diagram for an...Ch. 6.SE - What is the difference between a transition state...Ch. 6.SE - Prob. 20EDRMCh. 6.SE - Prob. 21EDRMCh. 6.SE - Draw an energy diagram for a two-step exergonic...Ch. 6.SE - Draw an energy diagram for a reaction with keq =...Ch. 6.SE - The addition of water to ethylene to yield ethanol...Ch. 6.SE - When isopropylidenecyclohexane is treated with...Ch. 6.SE - Prob. 26EDRMCh. 6.SE - Draw the electron-pushing mechanism for each...Ch. 6.SE - Draw the complete mechanism for each polar...Ch. 6.SE - Prob. 29EDRMCh. 6.SE - Identify the functional groups in the following...Ch. 6.SE - Identify the following reactions as additions,...Ch. 6.SE - Identify the likely electrophilic and nucleophilic...Ch. 6.SE - For each reaction below identify the electrophile...Ch. 6.SE - Prob. 34APCh. 6.SE - Follow the flow of electrons indicated by the...Ch. 6.SE - Prob. 36APCh. 6.SE - Prob. 37APCh. 6.SE - Despite the limitations of radical chlorination of...Ch. 6.SE - Prob. 39APCh. 6.SE - Answer question 6-39 taking all stereoisomers into...Ch. 6.SE - Prob. 41APCh. 6.SE - Prob. 42APCh. 6.SE - Prob. 43APCh. 6.SE - The reaction of hydroxide ion with chloromethane...Ch. 6.SE - Prob. 45APCh. 6.SE - Ammonia reacts with acetyl chloride (CH3COCl) to...Ch. 6.SE - The naturally occurring molecule α-terpineol is...Ch. 6.SE - Prob. 48APCh. 6.SE - Prob. 49APCh. 6.SE - Draw the structures of the two carbocation...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following statements islare true about fat soluble vitamins (A. D, E. & K)? (a) Fats are non-polar substance while Vitamin A, D. E and K are polar substances. (b) Both fats and Vitamins A. D. E, &K are non-polar substances. (c) These fat soluble vitamins if taken in excess can accumulate in the body fats. (d) Fats are needed by our body to dissolve these vitamins. a b and c a and d O b, c and d Which of the following statements is NOT true about intramolecular force? O It holds atoms together in a molecule. It is the chemical bond between atoms in a molecule. O It stabilizes the individual molecules O It is weaker than intermolecular force.arrow_forwardBy drawing the Lewis structure of the cyanide anion, interpret which atom the active tip is on (which atom it reacts on in a reaction) according to MOT?arrow_forwardHow many sigma and phi bonds are present in the amino acid glycine?arrow_forward
- The “free-base” form of cocaine (C17H21NO4) and its protonatedhydrochloride form (C17H22ClNO4) are shownbelow; the free-base form can be converted to the hydrochlorideform with one equivalent of HCl. For clarity, notall the carbon and hydrogen atoms are shown; each vertexrepresents a carbon atom with the appropriate number ofhydrogen atoms so that each carbon makes four bonds toother atoms.(a) One of these forms of cocaine is relatively water-soluble:which form, the free base or the hydrochloride?(b) One of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride? (c) The free-base form of cocaine has a solubility of 1.00 g in6.70 mL ethanol (CH3CH2OH). Calculate the molarityof a saturated solution of the free-base form of cocaine inethanol.(d) The hydrochloride form of cocaine has a solubility of1.00 g in 0.400 mL water. Calculate the molarity of a saturatedsolution of the hydrochloride form of cocaine inwater.(e) How many mL of a…arrow_forwardWrite an equation to show the reaction between ethanol, C2H5OH and methyllithium, CH3 Draw all non-bonding electrons and show electron flow with curved arrows.arrow_forwardLinoleic acid is an essential fatty acid found in many veg-etable oils, such as soy, peanut, and cottonseed. A key structural feature of the molecule is the c is orientation around its two dou-ble bonds, where R1and R2 represent two different groups thatform the rest of the molecule. (a) How many different compounds are possible, changing onlythe cis-trans arrangements around these two double bonds?(b) How many are possible for a similar compound with three double bonds?arrow_forward
- In each of the four molecules shown below the oxygen atom has two lone pairs of electrons to complete its Octet. H H H i H-C-C-H IT H-C-C-0-H || H HA H B H H H&CH -H HIC OIC-H ннс Η H D Which of these molecules could function as both a hydrogen bond donor and acceptor? OC and D none of the four molecules A and B all four of the molecules Aarrow_forwardDetergents need not be ionic. Pentaerythrityl palmitate (shown here) is a nonionic detergent used in dishwashingliquids.(a) Identify the hydrophilic and hydrophobic portions of the molecule.(b) Draw a depiction of a micelle that would form if this compound were dissolved in water.(c) What intermolecular interactions are primarily responsible for the micelle’s solubility in water?(d) What advantages do nonionic detergents have over ionic detergents in hard water?arrow_forwardIn the manufacture of margarine, unsaturated vegetable oils are converted to saturated fats by a process that converts C=C double bonds to C-C single bonds: Unsaturated fat Saturated fat What reagent may be used to accomplish this transformation? o hydrogen gas in palladium catalyst O dilute aqueous sulfuric acid O cold potassium permanganate in basic medium O room-temperature bromine waterarrow_forward
- Explain concisely why carbon has two electrons in different p orbitals with parallel spins rather than the other possible arrangement.arrow_forwardGiven that the condensed structure for GABA is H2N(CH2)3CO2H Find (A) the expanded structure and (B) the Line Structurearrow_forwardEncircle the most basic Nitrogen atomarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY