
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 95P
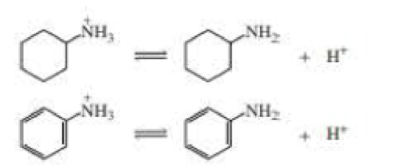
Protonated cyclohexylamine has a Ka = 1 × 10–11 Using the same sequence of steps as in Problem 94, determine which is a stronger base: cyclohexylamine or aniline.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Protonated cyclohexylamine has a Ka = 1 X 10-11, determine which is a stronger base: cyclohexylamine or aniline.
Rank the following conjugate bases in order of increasing basicity, putting the least basic first.
NH2
но
CH3
II
Multiple Choice
|| < |I| < |
|< || < |II
|| < |< |I|
|< ||| < ||
Which of the following has the lowest pka value? Hint: a methyl group is an
electron donating group which can destabilize the negative charge of the
conjugate base. *
I CH,COOH
I HCOOH
1 (CH),CH – COOH
II CH,CH,COH
IV
Chapter 8 Solutions
Organic Chemistry (8th Edition)
Ch. 8.1 - Prob. 1PCh. 8.1 - Prob. 2PCh. 8.4 - Prob. 3PCh. 8.5 - Prob. 4PCh. 8.5 - Prob. 6PCh. 8.6 - a. Predict the relative bond lengths of the three...Ch. 8.6 - Prob. 8PCh. 8.6 - Prob. 9PCh. 8.6 - Prob. 10PCh. 8.7 - Prob. 11P
Ch. 8.7 - Prob. 12PCh. 8.7 - Prob. 13PCh. 8.8 - Prob. 14PCh. 8.8 - Prob. 15PCh. 8.8 - Prob. 16PCh. 8.9 - Which member of each pair is the stronger acid?Ch. 8.9 - Which member of each pair is the stronger base? a....Ch. 8.9 - Rank the following compounds from strongest acid...Ch. 8.10 - Prob. 20PCh. 8.10 - Which acid in each of the following pairs is...Ch. 8.10 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.11 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - Prob. 28PCh. 8.12 - Prob. 29PCh. 8.12 - Prob. 30PCh. 8.12 - Prob. 31PCh. 8.12 - Prob. 32PCh. 8.13 - Prob. 33PCh. 8.13 - Prob. 34PCh. 8.13 - Prob. 35PCh. 8.13 - What are the major 1,2- and 1,4-addition products...Ch. 8.13 - Prob. 38PCh. 8.14 - Prob. 39PCh. 8.14 - Prob. 40PCh. 8.14 - Prob. 41PCh. 8.14 - Prob. 42PCh. 8.14 - Prob. 43PCh. 8.14 - Prob. 44PCh. 8.14 - Prob. 46PCh. 8.15 - Prob. 47PCh. 8.17 - Prob. 48PCh. 8.17 - Prob. 49PCh. 8.18 - Prob. 50PCh. 8.18 - Prob. 52PCh. 8.18 - Prob. 53PCh. 8.18 - Prob. 54PCh. 8.19 - Prob. 55PCh. 8.20 - Prob. 56PCh. 8.20 - What orbitals contain the electrons represented as...Ch. 8.20 - Prob. 59PCh. 8.20 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Which compound is the strongest base?Ch. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - a. The A ring (Section 3.16) of cortisone (a...Ch. 8 - Prob. 78PCh. 8 - Prob. 79PCh. 8 - Prob. 80PCh. 8 - Prob. 81PCh. 8 - Purine is a heterocyclic compound with four...Ch. 8 - Prob. 83PCh. 8 - Why is the delocalization energy of pyrrole (21...Ch. 8 - Prob. 85PCh. 8 - Prob. 86PCh. 8 - Prob. 87PCh. 8 - A student obtained two products from the reaction...Ch. 8 - Prob. 89PCh. 8 - a. How could each of the following compounds be...Ch. 8 - Draw the products obtained from the reaction of...Ch. 8 - How would the following substituents affect the...Ch. 8 - Prob. 93PCh. 8 - The acid dissociation constant (Ka) for loss of a...Ch. 8 - Protonated cyclohexylamine has a Ka = 1 1011...Ch. 8 - Draw the product or products that would be...Ch. 8 - Prob. 97PCh. 8 - Prob. 98PCh. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - a. Propose n mechanism for the following reaction:...Ch. 8 - Prob. 103PCh. 8 - As many as 18 different Diels-Alder products can...Ch. 8 - Prob. 105PCh. 8 - Prob. 106PCh. 8 - Prob. 107PCh. 8 - Prob. 108PCh. 8 - The experiment shown next and discussed in Section...Ch. 8 - Prob. 110PCh. 8 - Prob. 111PCh. 8 - Prob. 112PCh. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following compounds is most basic? OH | OH = || ||| NH₂ NH₂ IVarrow_forwardRank the following compounds in order of increasing acidity, putting the least acidic first. 6 CH3COOH FCH2COOH CICH2COOH BRCH2COOH I II III IV Multiple Choice | < IV < |I| < || |< ||| < IV < Il Il < |I| < IV < T Il < IV < |I| < |arrow_forwardWhich of the attached anions is the stronger base? Explain your choice.arrow_forward
- Using pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.arrow_forwardChoose the weak base from the compounds below. OCH3COOCH3 CH3SCH3 OCH3CH21 O(CH3)2NH OCH3CH3arrow_forwardWhat is the correct rank of the following compounds in order of decreasing acidity? 16 NH2 II IV Multiple Choice IV < III < || < | |< || < ||| < IV III < || < | < IV I| < IV < |< |Il|arrow_forward
- Which among the compounds below is the strongest base? II IV I IIIarrow_forwardRank the following organic molecules in order of decreasing acidity. That is, select '1' in the second column for the most acidic molecule, '2' for the next most acidic molecule, and so on. HO HO Jl. CI OH OH (Choose one) (Choose one) (Choose one) ▼ (Choose one) ▼ Xarrow_forwardRank the following in order of increasing basicity. The first in your list should be the least basic. HO 1 0 3 2 < 1 O2 3<1 01 2<3 01 3<2 3 1< 2 3. 2.arrow_forward
- Rank the following conjugate bases in order of decreasing basicity, putting the most basic first. H2C=CH HC=C CH3 II Multiple Choice || > |> |II |> || > III III > || > | III > | > ||arrow_forwardIndicate which is the weaker base and explain why. Be specific! NH3 or NF3arrow_forwardArrange the following compounds in order of decreasing acidity. I || > ||| > | O III >> || ||| > || > | O I > I| > ||| CH₂ II III 0arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY