
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 94P
The acid dissociation constant (Ka) for loss of a proton from cyclohexanol is 1 × 10–16
- a. Draw a reaction coordinate diagram for loss of a proton from cyclohexanol.

- b. Draw the resonance contributors for phenol.
- c. Draw the resonance contributors for the phenolate ion.
- d. On the same plot with the energy diagram for loss of a proton from cyclohexanol, draw an energy diagram for loss of a proton from phenol.
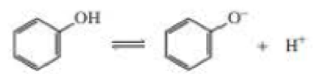
- e. Which has a greater Ka: cyclohexanol or phenol?
- f. Which is a stronger acid: cyclohexanol or phenol?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-
Which reaction will give a carboxylic acid?
O a. cyclopentanol + PCC
O b. tert-pentyl alcohol + CrO3/ H*
O c. 1-butanol + PCC
O d. 3-penten-1-ol + CrO3/ H*
e. 3-methyl-2-pentanol + Cro3/ H*
What is the best answer for the following reaction?
OH
Br
H.
A. 1) HO
OH H,o
3) H,o
2) Mg, Diethylether
B. 1) Mg, Diethylether 2) H30 3) HO
OH H,o*
с. 1) но
OH H3O 2) Mg, Diethylether
D. 1) Mg, Diethylether
2) HO
он
Chapter 8 Solutions
Organic Chemistry (8th Edition)
Ch. 8.1 - Prob. 1PCh. 8.1 - Prob. 2PCh. 8.4 - Prob. 3PCh. 8.5 - Prob. 4PCh. 8.5 - Prob. 6PCh. 8.6 - a. Predict the relative bond lengths of the three...Ch. 8.6 - Prob. 8PCh. 8.6 - Prob. 9PCh. 8.6 - Prob. 10PCh. 8.7 - Prob. 11P
Ch. 8.7 - Prob. 12PCh. 8.7 - Prob. 13PCh. 8.8 - Prob. 14PCh. 8.8 - Prob. 15PCh. 8.8 - Prob. 16PCh. 8.9 - Which member of each pair is the stronger acid?Ch. 8.9 - Which member of each pair is the stronger base? a....Ch. 8.9 - Rank the following compounds from strongest acid...Ch. 8.10 - Prob. 20PCh. 8.10 - Which acid in each of the following pairs is...Ch. 8.10 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.11 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - Prob. 28PCh. 8.12 - Prob. 29PCh. 8.12 - Prob. 30PCh. 8.12 - Prob. 31PCh. 8.12 - Prob. 32PCh. 8.13 - Prob. 33PCh. 8.13 - Prob. 34PCh. 8.13 - Prob. 35PCh. 8.13 - What are the major 1,2- and 1,4-addition products...Ch. 8.13 - Prob. 38PCh. 8.14 - Prob. 39PCh. 8.14 - Prob. 40PCh. 8.14 - Prob. 41PCh. 8.14 - Prob. 42PCh. 8.14 - Prob. 43PCh. 8.14 - Prob. 44PCh. 8.14 - Prob. 46PCh. 8.15 - Prob. 47PCh. 8.17 - Prob. 48PCh. 8.17 - Prob. 49PCh. 8.18 - Prob. 50PCh. 8.18 - Prob. 52PCh. 8.18 - Prob. 53PCh. 8.18 - Prob. 54PCh. 8.19 - Prob. 55PCh. 8.20 - Prob. 56PCh. 8.20 - What orbitals contain the electrons represented as...Ch. 8.20 - Prob. 59PCh. 8.20 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Which compound is the strongest base?Ch. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - a. The A ring (Section 3.16) of cortisone (a...Ch. 8 - Prob. 78PCh. 8 - Prob. 79PCh. 8 - Prob. 80PCh. 8 - Prob. 81PCh. 8 - Purine is a heterocyclic compound with four...Ch. 8 - Prob. 83PCh. 8 - Why is the delocalization energy of pyrrole (21...Ch. 8 - Prob. 85PCh. 8 - Prob. 86PCh. 8 - Prob. 87PCh. 8 - A student obtained two products from the reaction...Ch. 8 - Prob. 89PCh. 8 - a. How could each of the following compounds be...Ch. 8 - Draw the products obtained from the reaction of...Ch. 8 - How would the following substituents affect the...Ch. 8 - Prob. 93PCh. 8 - The acid dissociation constant (Ka) for loss of a...Ch. 8 - Protonated cyclohexylamine has a Ka = 1 1011...Ch. 8 - Draw the product or products that would be...Ch. 8 - Prob. 97PCh. 8 - Prob. 98PCh. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - a. Propose n mechanism for the following reaction:...Ch. 8 - Prob. 103PCh. 8 - As many as 18 different Diels-Alder products can...Ch. 8 - Prob. 105PCh. 8 - Prob. 106PCh. 8 - Prob. 107PCh. 8 - Prob. 108PCh. 8 - The experiment shown next and discussed in Section...Ch. 8 - Prob. 110PCh. 8 - Prob. 111PCh. 8 - Prob. 112PCh. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which member of each pair is the stronger base? a. ethylamine or aniline b. ethylamine or ethoxide ion c. phenolate ion or ethoxide ion d. phenolate ion or acetate ionarrow_forwardDraw the products of each acid-base reaction. EXAMPLE: a. b. CH3CH₂ propanoic acid OH || + CH3CH₂CH₂ OH + K+ OH This proton is transferred from the acid to the base. NaOH + base Na₂CO3 + H-O-H CH₂CH₂ O K+ The carboxylate anion is formed as a potassium salt.arrow_forwardExplain why the following reactions do not work Br OMs Br NaOH/H₂O Heat NaOH Acetone NaOH / H₂O Heatarrow_forward
- 2. Draw the products using curved arrows for the following acid base reaction. Label its conjugated acid-base pair. I—Z Base + N H. Acid H H searrow_forwardDraw the possible products of this epoxide ring-opening reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 41100 H 1) NaOH / H₂O 2) dilute HCI ☑Ⓒ Please select a drawing or reagent from the question areaarrow_forwardFor each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forward
- In each reaction, Label the reactants as a Lewis base (nucleophile) or a Lewis acid (electrophile). Use curved arrows to show the movement of electron pairs in the reaction. NH, H-C- H- H. ONH3arrow_forwardDraw the curved arrows and the products formed in the acid–base reaction of HBr and NH3. Determine the direction of equilibrium. Step 1: What happens in an acid–base reaction? Step 2: Draw the products of the acid–base reaction. Step 3: Draw the curved arrow mechanism of the acid–base reaction. Step 4: Determine the direction of equilibriumarrow_forwardleft 4. Consider the following equilibrium: 29925610 + Aga weak and Sung base Bast CORS + 1ght base: Strong ad d AddOH ress stable OH Is the carboxylate anion (CH3CH₂COO) an appropriate base to deprotonate the alcohol? A) yes, because the equilibrium favors the left side B) yes, because the equilibrium favors the right side C) no, because the equilibrium favors the left side D) no, because the equilibrium favors the right side OH COO basarrow_forward
- Choose the compound that is a ketotetrose. A B O A Ов Ос C OD H D О н-с-с-с-с-OH ОН Н Н H-C с- I- он н Н ОН ОН Н он н н н н H-C-С -с-с-о-Н ОН оно ОН ОН н н н н н H_C_C-с I-U ОН ОН ОН ОН О-Нarrow_forwardОН Type of reaction 1-Propanol is heated Type of reaction H+, Heat IZ + t 1- NaOHarrow_forwardDraw the major product for the following reaction. 1. NaOH H2O + H. 2. Dil H+arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY