
Interpretation: The reason behind specific product formed in below reaction should be written.
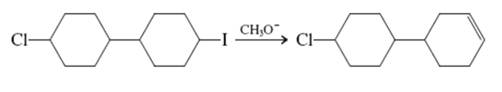
Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- Complete the following reactions by identifying the majority product(s) or the reaction conditions that are missing. No mechanism is neededarrow_forwardWhich of the reactions in has the least kinetically stable product?arrow_forwardPlease show the entire mechanism with curved arrows for the reaction. Why is the reaction considered unfavorable in reference to the stability ladder? Why does the reaction favor the product formation? *please show the resonance structures in the reaction.*arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
