
(a)
Interpretation: The products formed due to elimination and most likely mechanism for below reactionsshouldbe written.
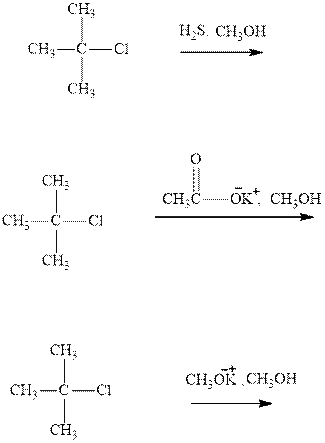
Concept introduction: Carbocations generated from
If the leaving group present eliminates along with a proton in absence of any strong nucleophile the double bond formation might occur. Such reactions are known as two step unimolecuar elimination, abbreviated as
(b)
Interpretation: The rates of below three reactions should be compared.
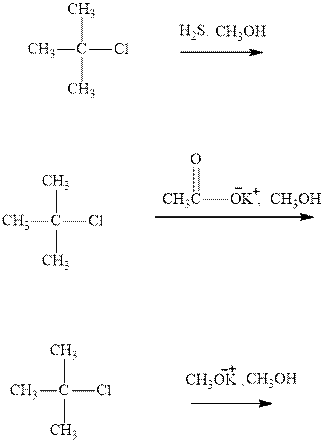
Concept introduction: Carbocations generated from alkyl halides have two fates; they can be either trapped by nucleophiles to give substitution product or may deprotonate to yield a small amount of alkene.
If the leaving group present eliminates along with a proton in absence of any strong nucleophile the double bond formation might occur. Such reactions are known as two step unimolecuar elimination, abbreviated as
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- 3. Complete the following reactions - draw the reactants and products, show the mechanism and conditions, and name the products. These reactions are one step. a) 2-Propanol + butanol → b) Ethanol + [O] → c) Butanone + [H] → d) 2-pentene + H₂O → e) Ethene + H₂ →→ f) Pentanol + HCI → g) Methanoic acid + 2-butanol → h) aminoethane + hexanoic acid → i) Benzene + chloroethane →→ j) Octane + O₂ → 2arrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forwardChlorination of 2-butanone yields two isomeric products, each having the molecular formula C4H7ClO. (a) What are these two compounds? (b) Write structural formulas for the enol intermediates that lead to each of these compounds. (c) Using curved arrows, show the flow of electrons in the reaction of each of the enols with Cl2.arrow_forward
- Butene (CH2CHCH2CH3) reacts with hydrogen chloride (HCl) at room temperature. Draw the displayed formulae of the products formed in this reaction. State the name of the above reaction mechanism. Alkene Propene reacts with hydrogen bromide (HBr) at room temperature to produce, 1-bromopropane and 2-bromopropane. H2C=CHCH3 + HBr → CH2(Br)CH2CH3 + CH3CH(Br)CH3 Draw out the reaction mechanism of the reaction between propene and hydrogen bromide to produce 2-bromopropane. Use curly arrows to show the movement of electrons. The description should be detailed and must include the type of bond fission that takes place. You may sketch and insert suitable diagrams to aid your description if you wish. Referring to the above mechanism, explain why two products are formed…arrow_forward5. a) Determine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. CH;CH,CH,CH,CCH, Br b) For the major elimination product obtained in 5a), which stereoisomer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forwardA certain hydrocarbon had the molecular formula C18H30 and contained two triple bonds. Ozonolysis gave CH₂(CH₂)CO₂H and HO₂CCH₂CH₂CO₂H as the only products. Draw a reasonable structure for this hydrocarbon. Click and drag to start drawing a structure.arrow_forward
- The rate of hydration of 2-butene and 2-methylpropene differ by several orders of magnitude. Please state which alkene is more rapidly hydrated and why.arrow_forward12) Use the curved arrow formalism to show the movement of electron pairs in the following reaction and label each reactant as a nucleophile or an electrophile. CHÍNH CHỊCH, + CO Học Nha CH₂CH₂CIarrow_forward5. a) Determine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. CH,CH,CH,CH,CCH, Br b) For the major elimination product obtained in 5a), which stereoisonmer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forward
- Which of the following is a chain propagation step in the free radical chlorination of methane? · CH3 + Cl · → CH3CI Cl2 2 Cl · CH4 + Cl HCl + · CH3 CH3 + CH4 CH4 + ·CH3arrow_forwardWhich of the following chemical equations depicts an alkylation reaction? C6H6() + CH3Cl() → C6H5CH3() + HCl(g) 2 CH3OH() + 3 O2(g) → 2 CO2(g) + 4 H2O() C6H12() → C6H10() + H2(g) CH2ClCH2Cl(g) + H2(g) → CH3CH3(g) + Cl2(g) CHClCHCl(g) → CH2ClCH2Cl(g)arrow_forwardRank the following alkenes in order of increasing rate of hydrogenation. 11 OI<|||< IV < II <|| < |||arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





