
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 26.SE, Problem 33AP
Interpretation Introduction
Interpretation:
The pKa1 and pKa2 of proline are 1.99 and 10.60 respectively. Using Henderson-Hasselbalch equation the ratio of protonated and neutral form at pH=2.5 and the ratio of neutral and deprotonated forms at pH=9.70 are to be calculated.
Concept introduction:
Henderson-Hasselbalch equation can be represented as
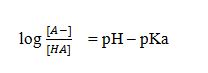
where [A-] is concentration of the ionized form and [HA] is the concentration of the unionized form at equilibrium.
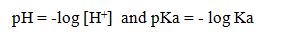
To calculate:
Using Henderson-Hasselbalch equation the ratio of protonated and neutral form at pH=2.5 and the ratio of neutral and deprotonated forms at pH=9.70 if the pKa1 and pKa2 of proline are 1.99 and 10.60 respectively.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Write the Henderson-Hasselbalch equation for a solution of methylamine. Calculate the quotient [CH3NH2]/[CH3NH13] at (a) pH 4.00; (b) pH 10.632; (c) pH 12.00.
You are looking at extracting a pyridine-bearing compound (conjugate acid pKA-4) from an organic phase and transfer it to the aqueous phase. Which of the following aqueous
solutions can you use to do so?
IM NGOH,pH 14
water, pH 7
saturated NH4Cl,pH 5
1MHCl, pH 1
AgNO3 + NaCl ->
Chapter 26 Solutions
Organic Chemistry
Ch. 26.1 - How many of the -amino acids shown in Table 26-1...Ch. 26.1 - Prob. 2PCh. 26.1 - Prob. 3PCh. 26.2 - Hemoglobin has pI=6.8. Does hemoglobin have a net...Ch. 26.3 - Show how you could prepare the following -amino...Ch. 26.3 - What alkyl halides would you use to prepare the...Ch. 26.3 - Prob. 7PCh. 26.4 - There are six isomeric tripeptides that contain...Ch. 26.4 - Draw the structure of M-P-V-G, and indicate its...Ch. 26.5 - Prob. 10P
Ch. 26.5 - Prob. 11PCh. 26.6 - The octapeptide angiotensin II has the sequence...Ch. 26.6 - Prob. 13PCh. 26.6 - Prob. 14PCh. 26.6 - Give the amino acid sequence of hexapeptides that...Ch. 26.7 - Prob. 16PCh. 26.7 - Write all five steps required for the synthesis of...Ch. 26.10 - Prob. 18PCh. 26.SE - Identify the following amino acids:Ch. 26.SE - Prob. 20VCCh. 26.SE - Isoleucine and threonine are the only two amino...Ch. 26.SE - Prob. 22VCCh. 26.SE - Give the sequence of the following tetrapeptide:Ch. 26.SE - Prob. 24MPCh. 26.SE - The chloromethylated polystyrene resin used for...Ch. 26.SE - Prob. 26MPCh. 26.SE - Prob. 27MPCh. 26.SE - Prob. 28MPCh. 26.SE - Except for cysteine, only S amino acids occur in...Ch. 26.SE - Prob. 30APCh. 26.SE - Prob. 31APCh. 26.SE - Show the structures of the following amino acids...Ch. 26.SE - Prob. 33APCh. 26.SE - Using both three- and one-letter codes for amino...Ch. 26.SE - Prob. 35APCh. 26.SE - Show how you could use the acetamidomalonate...Ch. 26.SE - Prob. 37APCh. 26.SE - Show how you could prepare the following amino...Ch. 26.SE - Prob. 39APCh. 26.SE - Predict the product of the reaction of valine with...Ch. 26.SE - Prob. 41APCh. 26.SE - Prob. 42APCh. 26.SE - Propose two structures for a tripeptide that gives...Ch. 26.SE - Show the steps involved in a synthesis of...Ch. 26.SE - Prob. 45APCh. 26.SE - Prob. 46APCh. 26.SE - Prob. 47APCh. 26.SE - Which of the following amino acids are more likely...Ch. 26.SE - Leuprolide is a synthetic nonapeptide used to...Ch. 26.SE - The -helical parts of myoglobin and other proteins...Ch. 26.SE - Prob. 51APCh. 26.SE - Cytochrome c is an enzyme found in the cells of...Ch. 26.SE - Prob. 53APCh. 26.SE - Prob. 54APCh. 26.SE - Prob. 55APCh. 26.SE - What is the structure of a nonapeptide that gives...Ch. 26.SE - Oxytocin, a nonapeptide hormone secreted by the...Ch. 26.SE - Aspartame, a nonnutritive sweetener marketed under...Ch. 26.SE - Prob. 59APCh. 26.SE - Prob. 60APCh. 26.SE - Prob. 61APCh. 26.SE - Prob. 62AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- You are going to be using Isoleucine in a buffer. The pKa of the carboxylate group of Isoleucine is 2.36 If you have a 0.1 M solution of Isoleucine at pH 3.22, what fraction (or percent) of the solution is in the deprotonated (COO- ) form?arrow_forwardCalculate the ratio of ethyl amine (CH3CH2NH2) to the ethyl ammonium ion (CH3CH2NH3+) in a solution with a pH of 11.0. pKa for CH3CH2NH3+ = 10.636.arrow_forwardAt pH 3.0, what percentage (in %) of lactic acid is in its deprotonated form? Hint: The pka of lactic acid is 3.86.arrow_forward
- Why do HCl, HNO3 etc. show acidic characters in aqueous solution while solutions of compounds like alcohol and glucose do not show acidic character?arrow_forwardIf a diprotic acid, H2A, has a pK1 = 2.20 and pK2 = 9.31, what is the principal species of this compound at pH = 5.13? O H2A O A2- OHA O H3A+arrow_forwardWhy do HCl, HNO3, etc. show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?arrow_forward
- 2. Will a 0.10 molar solution of CH3NH3OCN (methylamine cyanate) produce a basic or acidic solution. Explain quantitatively.arrow_forwardThe base methylamine ( CH3NH₂) has a Kb of 5.0 x 10-4. A closely related base, trimethylamine ( (CH3)3N), has a Kb of 7.4 x 10-5. a Which of the two bases is stronger? methylamine trimethylamine Correct Since methylamine has the larger K₁, methylamine is the stronger base. b Calculate the pH of a 0.38 M solution of the stronger base. pH =arrow_forwardIf 100 mls of 0.02 M HCl added to 400 mls of 0.5 M glycine buffer at pH10.4, what is the resultant pH? (pki for Gly= 2.2, Pk2=9.4) Part B: What would the pH be if 100 mls of .02 M HCl was added to 400 mls of water?arrow_forward
- Show the reaction and equilibrium expression for the basic dissociation of trimethylamine. Given that the Ka = 1.58 x 10-10 for trimethyl ammonium ion, use this to calculate the equilibrium constant for the basic dissociation of trimethyl amine. If the concentration of trimethylamine is 0.10 M, what is the pH of the solution?arrow_forwardWill 3- methylhexanoic acid be negative or neutral at pH of 6.38?arrow_forwardWhat is the ratio of lactic acid (Ka = 1.37x10-4) to lactate in a solution with pH =4.09? Write your ratio out as a number containing a decimal. [lactic acid][lactate] =arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
