
Concept explainers
Interpretation:
The
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
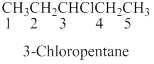
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
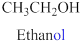

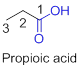
The given compound is an acid (
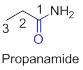
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
If the molecule is ester,
Esters end with “ate”
Example
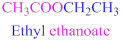
The given compound is an
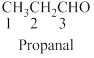
The given compound is
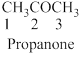
The given compound is an
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry: Atoms First
- Hello, can you draw and name two different molecules (with the explicit formula) with a total of 12 carbons, together with the side groups, according to the options given? a)Write and name two cyclic amide molecule. b)Write and name two aromatic amine molecule.arrow_forwardHello, can you draw and name two different molecules (with the explicit formula) with a total of 12 carbons, together with the side groups, according to the options given? a)Write and name two beta-dicarbonyl molecules. b)Write and name two quaternary ammonium salts. c)Write and name two tertiary amine molecules.arrow_forwardWhich of the following functional groups would convert a hydrocarbon into a hydrophilic molecule. Select all that apply. Carboxylic acid Amine group Hydroxyl group Methyl grouparrow_forward
- Identify which of the following statement(s) is/are true. (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one -OH group, a secondary alcohol has two -OH groups, and a tertiary alcohol has three -OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N.arrow_forward. (a) Write the molecular formula of acetylsalicylic acid (see Fig. 7.34a).(b) An aspirin tablet contains 325 mg acetylsalicylic acid. Calculate the number of moles of that compound in the tablet.arrow_forwardWhat is the complete IUPAC name for each of the following molecules? (a) Br (b) CI (c) F (d) Ph OCH3 CIarrow_forward
- Write down the distinguishing features among the following: (a) primary,secondary,tertiary Alcohols (b)primary, secondary, tertiary amines (c) carboxylic acid,ester and amide (d)Alkenes Alkynesarrow_forwardWhich of the following is correctly paired? Choose all that apply Carboxylic acid: oxygen Thiol: phosphorus Phosphate: Oxygen Amine: Nitrogenarrow_forwardDetermine the IUPAC name of each of the following molecules. (a) Br (b) (c) CI (d) (e) H3CO NO,arrow_forward
- Carbon bonds to many elements other than itself.(a) Name six elements that commonly bond to carbon in organiccompounds.(b) Which of these elements are heteroatoms?(c) Which of these elements are more electronegative than car-bon? Less electronegative?(d) How does bonding of carbon to heteroatoms increase thenumber of organic compounds?arrow_forward(b) Draw the structural formula for each of the following compounds. Lukiskan formula struktur bagi setiap sebatian berikut. (i) 2-iodo-4-methylpentan-3-amine (ii) 1,1-dibromo-2-chloro-2-methylbutane (iii) 2-ethyl-6-fluorophenolarrow_forwardDraw structures for the following compounds. Show all of the hydrogen atoms in the molecules. (d) ethanoic acid (e) 1,4-dichlorobenzene (f) 2-butanonearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





