
Concept explainers
Interpretation:
The nitrogen containing
Concept introduction:
If the compound is having oxygen atom it must be an alcohol
Example is given below
If any halogens are present in the molecule.
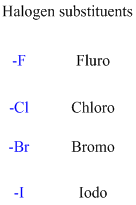
If the molecule is having hydroxyl group is called as an alcohol.
For example:
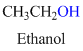
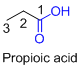
If the molecule is having (
For example:
If the molecule is having (
For example
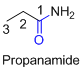
If the molecule is having (
For example

If the molecule is having (
Example
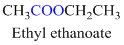
If the molecule is having (
Example

The given compound is (
Example

Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry: Atoms First
- Draw the structures of the following compounds: N-ethyl-N-methylhexan-3-aminearrow_forwardAmoxicillin, an antibiotic, has the following structure. Identify the functional groups in amoxicillin. NH2 H. -N- но HOarrow_forwardIdentify which of the following statement(s) is/are true. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forward
- Draw and Name FIVE constitutional isomers C4H8O2 Ester or Carboxylic Acidarrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forwardThe ester with the formula C8H16O2 gives an alcohol and an acid when hydrolyzed. When the alcohol is isolated and oxidized, it forms a ketone. Which of these formulas cannot be the ester?arrow_forward
- what is the semi structural formula and molecular formula of pentan-2-amine, 2-chloropan-1-ol, 2hydroxypropanoic acid. Molecule and structural formula if ch3chclch2clarrow_forwardDraw three isomers of C5H10O2C5H10O2. At least one should contain an an ester, one should contain a carboxylic acid, and one should contain an etherarrow_forwardMany naturally occurring compounds contain more than one functional group. Identify the functional groups in the following compounds:(a) Penicillin G is a naturally occurring antibiotic.(b) Dopamine is the neurotransmitter that is deficient in Parkinson’s disease.(c) Capsaicin gives the fiery taste to chili peppers.(d) Thyroxine is the principal thyroid hormone.(e) Testosterone is a male sex hormone.arrow_forward
- You were tasked to propose a structure for a compound with a chemical formula of C₂H₂N. In the laboratory, the compound appears as a clear, colorless liquid. To determine the structure of the compound, you subject the compound to several solubility tests and chemical tests. The observations for these tests are summarized: SOLUBILITY GROUP: Group V Insoluble in water and 10% NaOH but soluble in 10% HCI FUNCTIONAL GROUP/CLASS: Amine 1) Based on the results of the solubility tests and chemical tests, what is the most probable structure of C7H9N? Draw your answer in the box below. You may draw the compound using line-bond formula OR Lewis structure. |arrow_forwardThe following compound is classified as a(n) alcohol O aldehyde amide O ether alkyne O anhydride ester O carboxylic acid O ketone aromatic O aminearrow_forwardVanillin, C8H8O3, is the active ingredient in vanilla flavoring. It contains a six-membered aromatic ring with an aldehyde group on carbon 1, an alkoxy group (ether) on carbon 3, and a hydroxyl group (alcohol) on carbon 4. Draw the structure of vanillin.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


