
Concept explainers
(a)
Interpretation:
A concept map is to be drawn and the grams of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 16E
The concept map is shown below.
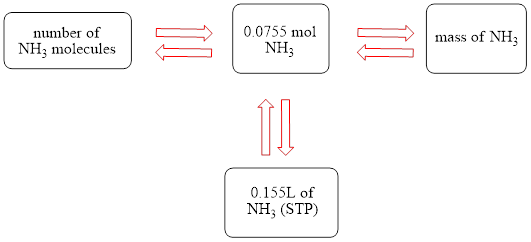
The grams of
Explanation of Solution
When

Figure 1
The molar mass of
Therefore, the mass of
The formula to calculate the mass of
Substitute the mass of
Therefore, the grams of
The grams of
(b)
Interpretation:
A concept map is to be drawn and the liters of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 16E
The concept map is shown below.

The liters of
Explanation of Solution
When

Figure 1
The volume occupied by
The formula to calculate the volume occupied by
Substitute the volume of
Therefore, the liters of
The liters of
(c)
Interpretation:
A concept map is to be drawn and the molecules of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 16E
The concept map is shown below.

The molecules of
Explanation of Solution
When

Figure 1
The molecules present in
The formula to calculate the molecules occupied by
Substitute the molecules in
Therefore, the molecules of
The molecules of
(d)
Interpretation:
A concept map is to be drawn and the molar concentration of the ammonia solution when
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 16E
The concept map is shown below.

The molar concentration of the ammonia solution is
Explanation of Solution
When

Figure 1
The number of moles in
The formula to determine molarity is shown below.
Where
•
•
•
Substitute the value of number of moles and volume in equation (1).
The relation between
The unit factors are given below.
The unit factor to determine
Therefore,
Therefore, the molar concentration of
The molar concentration of
Want to see more full solutions like this?
Chapter 15 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- If .30 mol of CuCO3 dissolved in 120 ml of water, what is the molarity of the solution?arrow_forwardHow many milliliters of 0.372 M NH4Cl will contain the following? (answer in scientific notation) (a) 0.52 mol NH4Cl (b) 31.8 g NH4Cl (c) 1.05 x 1020 formula units of NH4Clarrow_forwardWhat percent of water is in the compound barium chloride dihydrate?arrow_forward
- What will be the effect on the calculated percent water in the unknown (greater or smaller), if the following events occur? Explain your answers.(a) Some of the hydrate spatters out of the crucible.(b) The crucible containing the hydrate is not heated long enough.(c) The empty covered crucible is weighed while it is hot.arrow_forwardIf 33.8 mL of an lead (II) nitrate solution is needed to precipitate all the chloride ions in a 836 mg sample of potassium chloride (forming lead (II) chloride), what is the molarity of the lead (II) nitrate solution? (Solution stoichiometry)arrow_forwardMolarity of solutionarrow_forward
- Refer to the oxygen concentration example in Sec. 1.7.2.(a) Given that nitrogen is lighter in weight than oxygen, is N2 concentration at 10 kmmore or less that 25% of the sea level N2 concentration?(b) What is the ratio of N2 concentration to O2 at 10 km? At sea level, the ratio is 4 to 1.arrow_forwardIn a dilute solution of sodium chloride in water, the sodium chloride is the: a. solvent. b. solute. c. precipitate. d. reactant.arrow_forwardPotassium hydroxide is used in making liquid soap. How many grams would you use to prepare 2.50L of 1.40M potassium hydroxide?arrow_forward
- what is the mass of a hydrated salt?arrow_forwardCauclate number of moles of waterarrow_forwardCalculate the number of grams of solute in each of the following solutions. (a) 4.86 L of a 2.56 M HCl solution grams of HCl (b) 44.6 mL of a 12.9 M HNO3 solution grams of HNO3 (c) 60.2 mL of a 10.7 M AgNO3 solution grams of AgNO3 (d) 1.85 L of a 5.6×10-3 M Na2SO4 solution grams of Na2SO4arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





