
Concept explainers
a.
Interpretation:
The compound
Concept Introduction:
Ammonium salts are similar to ionic compounds. They have cation and anion. Ammonium salts are highly soluble in water.
b.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
d.
Interpretation:
The given compound is water soluble or water insoluble has to be labelled.
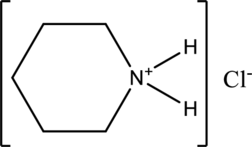
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the possible products of this epoxide ring-opening reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 41100 H 1) NaOH / H₂O 2) dilute HCI ☑Ⓒ Please select a drawing or reagent from the question areaarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. I I I I Mg. :O: Br. :O: Select to Add Arrows :O: CO2 THE H Mg 0: Br Please select a drawing or reagent from the question areaarrow_forwarda) Rank the following in order of increasing boiling point. HO CH3 A B b) Rank the following in order of increasing acidity. H3C `OH HS. но. `OH HO Aarrow_forward
- Show how to bring about each conversion in good yield. a. b. C6H5 Cl OH COOH C6H5 COOHarrow_forwardDraw the products of each acid-base reaction. H CH3 H CH,CH,NHCH3 COOH + а. NAOH b. CF3 + HCI CH,O naproxen anti-inflammatory agent fluoxetine antidepressant 1 1arrow_forwardDraw the products of each acid-base reaction. NaOH b. + NazCO3 a. + CH3CH,CH, HO, OHarrow_forward
- Which is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-arrow_forward4. Which of the following is the strongest base? A. (CH3)3COH B. CH3CH₂NH™ C. H₂O D. CH3NH2arrow_forwardRank the below compounds in order of increased acidity and explain your choice. HO. H3C || HO. HO.arrow_forward
- What is the structure of compound A? 010 Compound A tot 01 Oll O III O IV OV || s ||| Compound A IV COOHarrow_forwardDraw the products of each acid-base reaction. NaOH а. HO, NaOCH3 b. OH С. NaH ОНarrow_forwardOH 15. CH3-C-H ÓCH, The compound above is an example of a(n) a. acetal b. ketal O c. hemiacetal d. hemiketal e. none of the abovearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
