
Concept explainers
What
or
a. ![Chapter 12, Problem 12.44P, What alkene is needed to synthesize each 1,2-diol using the [1] OsO4 followed by NaHSO3 in H2O; or , example 1](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9780078021558/Chapter-12/images/21558-12-12.44p-question-digital_image001.jpg) b.
b. ![Chapter 12, Problem 12.44P, What alkene is needed to synthesize each 1,2-diol using the [1] OsO4 followed by NaHSO3 in H2O; or , example 2](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9780078021558/Chapter-12/images/21558-12-12.44p-question-digital_image002.jpg) c.
c. ![Chapter 12, Problem 12.44P, What alkene is needed to synthesize each 1,2-diol using the [1] OsO4 followed by NaHSO3 in H2O; or , example 3](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9780078021558/Chapter-12/images/21558-12-12.44p-question-digital_image003.jpg)
(a)
Interpretation: The alkene needed to synthesize the given
Concept introduction: Addition of two hydroxyl groups on double bond to form
In the presence of peroxide, alkene is oxidized to epoxide. This is known as epoxidation. This is a syn addition. The weak pi bond of alkene and weak
Answer to Problem 12.44P
The alkene needed to synthesize the given
Explanation of Solution
In presence of
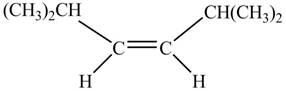
Figure 1
The corresponding chemical reaction is given below.
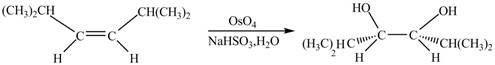
Figure 2
In presence of
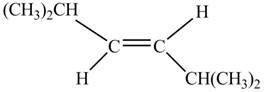
Figure 3
The corresponding chemical reaction is given below.
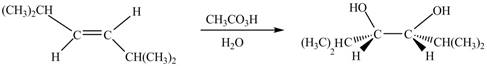
Figure 4
The alkene needed to synthesize the given
(b)
Interpretation: The alkene needed to synthesize the given
Concept introduction: Addition of two hydroxyl groups on double bond to form
In the presence of peroxide, alkene is oxidized to epoxide. This is known as epoxidation. This is a syn addition. The weak pi bond of alkene and weak
Answer to Problem 12.44P
The alkene needed to synthesize the given
Explanation of Solution
In presence of
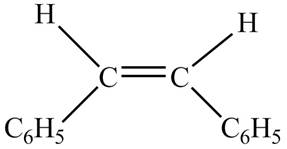
Figure 5
The chemical reaction is given below.
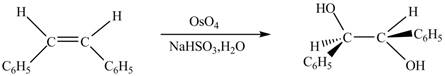
Figure 6
In presence of
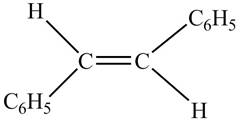
Figure 7
The chemical reaction is given below.
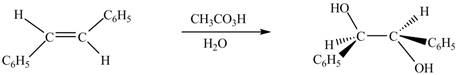
Figure 8
The alkene needed to synthesize the given
(c)
Interpretation: The alkene needed to synthesize the given
Concept introduction: Addition of two hydroxyl groups on double bond to form
In the presence of peroxide, alkene is oxidized to epoxide. This is known as epoxidation. This is a syn addition. The weak pi bond of alkene and weak
Answer to Problem 12.44P
The alkene needed to synthesize the given
Explanation of Solution
The given alkene is,
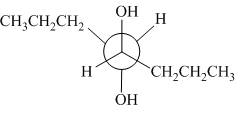
Figure 9
This is a staggered conformation in which two hydroxyl groups are anti to each other.
In presence of
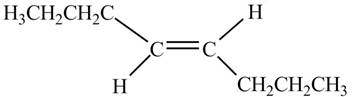
Figure 10
The chemical reaction is given below.
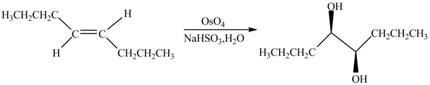
Figure 11
In presence of
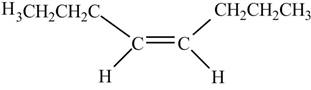
Figure 12
The chemical reaction is given below.
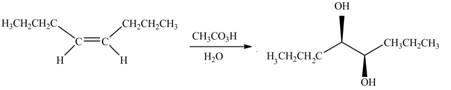
Figure 13
The alkene needed to synthesize the given
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry (7th Edition)
Organic Chemistry - Standalone book
Introduction to Chemistry
Basic Chemistry (5th Edition)
Principles of General, Organic, Biological Chemistry
- OH conc H3PO4 A A MCPBA CH₂Cl₂ B NaOH H₂O 8. Identify the products A,B and C.arrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardWhich is the major organic product of this reaction? A. B. 4 ОА O OB O C COD AICI3 benzene C. D.arrow_forward
- Draw the structure of the organic product or products formed in the reaction. H₂C HU IN. H HU of H₂ MgBr 1. H₂C A -CH₂ 2. H3O+ Select Draw / || III Rings C H Morearrow_forwardDetermine which reactions would occur (substitution, elimination, both, or neither). CH;CH,OH a. SN1 O b. E1 C. SN1 & E1 O d. SN2 е. Е2 O f. SN2 & E2 g. No reactionarrow_forwardWhich reagent(s) convert a carbonyl group (C=O) into a methylene group (CH2)? O A. Zn / HCI O B. LIAIH4 / Et20 O C. NABH4 / MeOH D. Na in liq. NH3arrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forwardPrepare the following compounds using cyclohexane as the starting material OH COOH b. „CH;OH CH,NH2 d. CH;CH,OH „CHCH, a.arrow_forward
- What alkene is needed to synthesize each 1,2-diol using [1] OsO4 followed by NaHSO3 in H2O; or [2] CH3CO3H followed by −OH in H2O?arrow_forwardWhat is the major organic product obtained from the following reaction? A. B. Yo OH OH 1. LiAlH4 2. H3O+ C. D. H OHarrow_forward6. Which of the following alcohols is oxidized to a ketone by chromic acid? I. II. III. I. II. OH III. OH CH₂OH 7. What are the major organic products when the following molecule is treated with ozone, and then with Zn/H2O? CH₂ Yu A. I B. II C. III D. IV E. V ,COH O IV. H-C-H IV. COOH V. 2 CH₂O CH₂OH CH₂ C-CH3 OH V. -OH Jº. n&on но- -он Y. A. I B.II C.III D.IV E. V 8. What compound results when 1-heptyne undergoes ozonolysis? A. heptanal B. 2-heptanone C. heptanoic acid D. hexanoic acid and CO2 E. 1-heptanolarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





