
a)
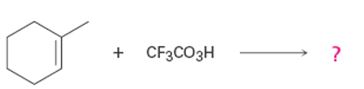
Interpretation:
Product formed in the reaction with the mechanism of its formation is to be given.
Concept introduction:
To give:
Product formed in the reaction with the mechanism of its formation.
b)
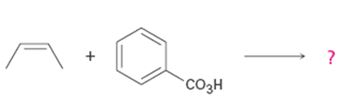
Interpretation:
Product formed in the reaction with the mechanism of itsformation is to be given.
Concept introduction:
Alkenes are oxidized to give epoxides on treatment with peroxyacids. The addition of oxygen to the double bond occurs with syn stereochemistry- both C-O bonds are formed on the same face of the double bond. The oxygen atom farthest from the carbonyl group is transferred to the double bond.
To give:
Product formed in the reaction with the mechanism of its formation.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- III. Give a detailed mechanism for the following reaction: OH ОНarrow_forwardGive the major products or reagents represented by letters E and F:arrow_forwardPredict the products of conjugate (Michael) additions, and show how to use thesereactions in syntheses. Show the general mechanism of the Robinson annulation,and use it to form cyclohexenone ring systems.arrow_forward
- Propose a mechanism to account for the products formed in the following reaction: The peroxide works as light, hν. So, read “NBS/hν.”arrow_forwardWhat is the synthesis and mechanism for the following?arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
- Complete the reaction schemes below providing the reagents required to achieve thetransformation. More than one step may be necessary for each scheme.arrow_forwardPropose a mechanism for the conjugate addition of a nucleophile (Nuc:-) to acrylonitrile(H2C“CHCN) and to nitroethylene. Use resonance forms to show how the cyano andnitro groups activate the double bond toward conjugate addition.arrow_forwardStep 4: In an acid-catalyzed epoxide ring opening, the epoxide is first protonated and can then be attacked by weak nucleophiles. H HO: :Nuc Nuc Weak nucleophiles such as water and alcohols can react with the protonated epoxide. One of the main differences between base-catalyzed and acid-catalyzed epoxide ring openings is the regiochemical product expected. In an acid-catalyzed reaction, if one of the epoxide carbons is tertiary and the other primary, the nucleophile will attack at the more substituted carbon. If the epoxide is monosubstituted, the nucleophile will attack preferentially at the primary carbon vs. the secondary carbon. Which reaction will not give the indicated product? N 1. CH,CH,O 2. H₂O OH CH₂CH₂OH, H* N CH₂CH₂OH, H 1 CHICH,O 2. H₂O HO HO H- Harrow_forward
- Provide the mechanism for the following reaction (1) PhMgBr (2 equiv) (2) H3O+ workup OHarrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forwardTreatment of 1,2-dibromoethane with the dithiolate dianion shown in the reaction below leads to two products as shown. Draw the structure for each molecular formula and provide a detailed mechanism for the formation of both products.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

