
Interpretation:
Names and chemical formulas for any five acids have to be given and any three observable properties generally associated with acids have to be listed.
Concept Introduction:
Strong Acids: Acids that dissociates into ions completely which results in easy donation of protons are considered as strong acids. Strong acid forms weaker conjugated base.
Weak Acids: Acids that do not easily dissociate into ions completely which has difficulty in proton donation are considered as weak acids. Weak acid forms stronger conjugated base
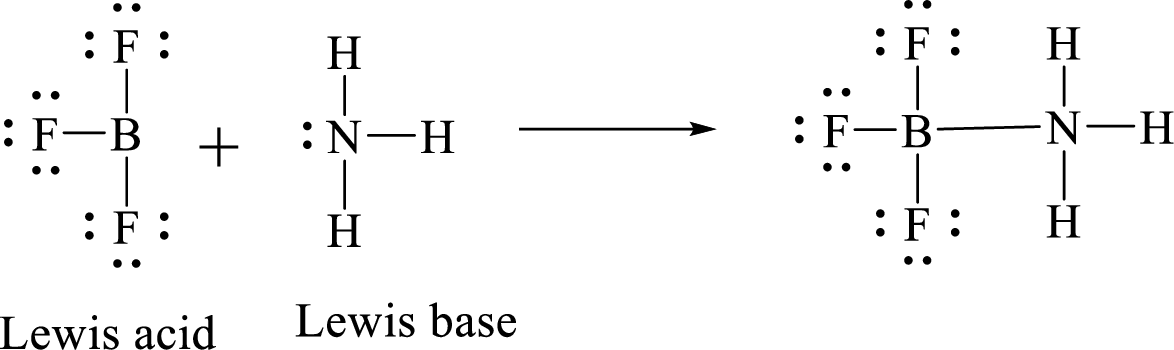
According to Lewis concept of acid and bases,
Lewis acid is an electron pair acceptor.
Lewis base is an electron pair donor.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Chemistry In Context
- Consider the salts listed in the left-hand column of the table. Determine the formula of the acid and base the salt originated from. Determine whether the salt is acidic, basie, or neutral.arrow_forwardlithium hydrogen sulfite Express your answer as a chemical formula. ΑΣφ A chemical reaction does not occur for this question. Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Part B sodium permanganate Express your answer as a chemical formula. ?arrow_forwardArrange the following sets of compounds based on the property given in increasing order. Type in the number of the compound to the corresponding roman numeral, being the lowest and IV being the highest. ACIDITY COOH COOH .COOH .COOHarrow_forward
- Use the information page to give the acid name and the acid formula for each of the ate anions below. Anion Name Anion Formula perchlorate CIO4 sulfate nitrate 2- SO4²- NO3 Acid Name Acid Formula Are the molecular compounds strong acids or weak acids in aqueous solution?arrow_forwardThe chemical formula for water is H2O. What is the chemical formula for methane?arrow_forwardWrite balanced complete ionic equation for CaS(aq) +CdCl2 (aq) → CdS(s) + CaCl (aq). Express your answer as a chemical equation. Identify all of the phases in your answer. ΑΣφ You have already submitted thinarrow_forward
- e 2N2(g) +502 (g) → 2N2O5 (g) BeCl2 (s) + H₂O(g) → BeO(s) + HCl(g) BeCl₂ (s) + H₂O(g) ->> BeO(s) + BaH₂ (s) + H₂O(l) → Ba(OH)2 (s) + H₂(g) BaH₂ (s) + H₂O(l) Submit Answer - Ba(OH)2 (s) + Retry Entire Group 1 more group attempt remaining Cengage Learning Cengage Technical Support HC1(g) H₂(g)arrow_forwardHere are the formulas for each substance: Sucrose: C12H22O11C_{12}H_{22}O_{11}C12H22O11 Copper (I) Chloride: CuClCuClCuCl Iodine: I2I_2I2 Potassium Iodide: KIKIKI Our goal is to try to find a pattern for which compounds conduct electricity when dissolved in water, and which don't. One hint is to look for patterns in which kinds of atoms form substances that conduct when dissolved, and which kinds of atoms form substances that don't conduct when dissolved. Here is a periodic table you can use. Let's start by finding the location on the periodic table for the atoms for substances that conduct when dissolved. What do you notice about where the atoms are?arrow_forwardWhat is the chemical opposite of an acid?arrow_forward
- Phosphoric acid, H3PO4, has K1 - 7.5 x 10 3 Ka2 = 6.3x 10 8 K3= 4.7x 10 13 HPO42 + H20 = HPO4* + OH K = ? O 2.1x 103 O 1.3x10 13 O 1.6 x 107 O 6.3x 108 O 4.7x 10 13arrow_forwardHow do you complete this table correctly?arrow_forward"What is the name of the ion with the formula Sn2+? Do not put the word ""ion"" in your answer. OH- would be entered as hydroxide." "What is the name of the ion with the formula NO3-? Do not put the word ""ion"" in your answer. OH- would be entered as hydroxide." "What is the name of the ion with the formula HCO 3 -? Do not put the word ""ion"" in your answer. OH - would be entered as hydroxide." "What is the name of the ion with the formula Mg2+? Do not put the word ""ion"" in your answer. OH- would be entered as hydroxide."arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning




