
(a)
Interpretation:
The given pairs which one is take place more rapid reaction have to be identified.
Concept Introduction:
Nucleophile is a chemical species that gives an electron pair to an electrophile to form a
Charged nucleophile is stronger than neutral nucleophiles.
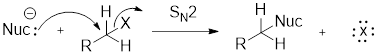
Structure of the substrate plays major role in the reactivity of
(b)
Interpretation:
The given pairs which one is take place more rapid reaction have to be identified.
Concept Introduction:
Nucleophile is a chemical species that gives an electron pair to an electrophile to form a chemical bond in relation to a reaction.
Charged nucleophile is stronger than neutral nucleophiles.
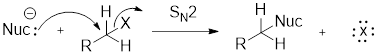
Structure of the substrate plays major role in the reactivity of
(c)
Interpretation:
The given pairs which one is take place more rapid reaction have to be identified.
Concept Introduction:
Nucleophile is a chemical species that gives an electron pair to an electrophile to form a chemical bond in relation to a reaction.
Charged nucleophile is stronger than neutral nucleophiles.
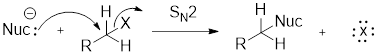
Structure of the substrate plays major role in the reactivity of
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Essential Organic Chemistry (3rd Edition)
- Which will react faster??arrow_forward2) Use your understanding of reaction rates to identify the faster reaction for each of the following pairings. HC Br O CECH Br 2 equiv of OR H NH2 Br 2 equiv of ONH2 Brarrow_forwardWhich of the following reactions take place more rapidly when the concentration of the nucleophile is increasedarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
