
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22.SE, Problem 57AP
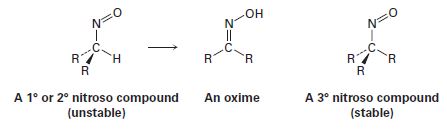
All attempts to isolate primary and secondary nitroso compounds result solely in the formation of oximes. Tertiary nitroso compounds, however, are stable. Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
A task is assigned to an undergraduate student to test two samples (known as
compounds K and L) in the laboratory. She placed these two compounds through
various scientific tests. She discovered that these compounds have the same
molecular formula, CSHSO. When treated with 2,4-dinitrophenylhydrazine, all of
these compounds produce brightly coloured precipitate, and both are reduced to an
organic compound with the molecular formula C§H100. However, compound K can
be easily oxidized by chromic acid to formed compound N and vice versa for
compound L. Furthermore, when both compounds react with Fehling's solutions,
they produce negative results. However, only compound K forms a silver mirror
when it reacts with Tollen's reagent, and compound L does not.
Identify the possible structural formulae for compounds K, L, and N by ignoring
their position isomerism.
Indicate the formation of compound N from compound K.
Predict the chemical reaction that occurs when compound L reacts with 2,4-…
3. It is required to introduce a halogen group to a five membered ring, thiophene. Discuss
the reaction mechanism involved in the reaction by selecting a suitable halogen group and
analyze why a particular substituted product obtained after the reaction is predominant
over the other possible product(s) with the help of reactions.
To meet the ever increasing demand of global population, the demand for textile products and
consumption of dyes by these industries is increasing.
Benzene is often produced as a side product during Grignard reactions using phenylmagnesium bromide. How can its formation be explained? Give a balanced equation for its formation.
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 1PCh. 22.1 - How many acidic hydrogens does each of the...Ch. 22.1 - Prob. 3PCh. 22.3 - Write the complete mechanism for the deuteration...Ch. 22.3 - Prob. 5PCh. 22.4 - If methanol rather than water is added at the end...Ch. 22.5 - Prob. 7PCh. 22.5 - Draw a resonance structure of the acetonitrile...Ch. 22.6 - If methanol rather than water is added at the end...Ch. 22.7 - Prob. 10P
Ch. 22.7 - Draw a resonance structure of the acetonitrile...Ch. 22.7 - Why do you suppose ketone halogenations in acidic...Ch. 22.7 - Prob. 13PCh. 22.7 - Prob. 14PCh. 22.7 - Prob. 15PCh. 22.7 - Prob. 16PCh. 22.SE - Prob. 17VCCh. 22.SE - Prob. 18VCCh. 22.SE - Prob. 19VCCh. 22.SE - Prob. 20MPCh. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Prob. 23MPCh. 22.SE - In the Hell–Volhard–Zelinskii reaction, only a...Ch. 22.SE - Prob. 25MPCh. 22.SE - Nonconjugated , -unsaturated ketones, such as...Ch. 22.SE - Prob. 27MPCh. 22.SE - Using curved arrows, propose a mechanism for the...Ch. 22.SE - Prob. 29MPCh. 22.SE - One of the later steps in glucose biosynthesis is...Ch. 22.SE - The Favorskii reaction involves treatment of an...Ch. 22.SE - Treatment of a cyclic ketone with diazomethane is...Ch. 22.SE - Prob. 33MPCh. 22.SE - Amino acids can be prepared by reaction of alkyl...Ch. 22.SE - Amino acids can also be prepared by a two-step...Ch. 22.SE - Heating carvone with aqueous sulfuric acid...Ch. 22.SE - Identify all the acidic hydrogens (pKa 25) in the...Ch. 22.SE - Rank the following compounds in order of...Ch. 22.SE - Prob. 39APCh. 22.SE - Base treatment of the following , -unsaturated...Ch. 22.SE - Prob. 41APCh. 22.SE - Prob. 42APCh. 22.SE - Prob. 43APCh. 22.SE - Which, if any, of the following compounds can be...Ch. 22.SE - Prob. 45APCh. 22.SE - Prob. 46APCh. 22.SE - Prob. 47APCh. 22.SE - How might you convert geraniol into either ethyl...Ch. 22.SE - Prob. 49APCh. 22.SE - One way to determine the number of acidic...Ch. 22.SE - Prob. 51APCh. 22.SE - Prob. 52APCh. 22.SE - Prob. 53APCh. 22.SE - Prob. 54APCh. 22.SE - Prob. 55APCh. 22.SE - Prob. 56APCh. 22.SE - All attempts to isolate primary and secondary...Ch. 22.SE - How would you synthesize the following compounds...Ch. 22.SE - Prob. 59APCh. 22.SE - Prob. 60APCh. 22.SE - Prob. 61APCh. 22.SE - Prob. 62APCh. 22.SE - As far back as the 16th century, South American...Ch. 22.SE - The key step in a reported laboratory synthesis of...Ch. 22.SE - Prob. 65AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the major products for the reaction. Cl₂ (low conc.)arrow_forwardprovide appropriate names for the following moleculesarrow_forward3. cyclohexanone to cyclohexane. A second year chemistry student is assigned a task to prepare compound 2 from compound 1 using lithium aluminium hydride as a reducing agent. However, from the reaction mixture, no traces of product 2 were detected. Explain this observation in detail and suggest an alternative route that will lead to product 2. H 1 CH3 LiAllH4 H O: 2 CH3 quearrow_forward
- Modify the structure of the compound provided to show the skeletal structure of the product that forms when this compound is treated with K₂Cr₂O. ☐ No reaction occurs. X :0 Garrow_forwardX Upon ozonolysis, Compound X produces two compounds: Compound Y and Compound Z. Compound Y can also be prepared from the following synthetic route: PCC 1. R₂BH, THF 1. Mg. Et₂O PCC Compound Y 2. CH₂Cl₂ 2. NaOH, HO CH₂Cl₂ 3. H₂O* From this information, draw the structures of Compounds X, Y, and Z. For Compounds X and Z, different substituents are possible. For grading purposes, just use hydrogens as the substituents. Br مرد →] ►arrow_forwardgive a primary organic product(s) for each reaction shownarrow_forward
- 1. In the reactions involving the three isomeric alcohols with the formula C4H9OH, describewhat each of the following tests showed about reactivity of the -OH group and reactions of 1°,2°, and 3° alcohols.• the test with neutral KMnO4• the test with concentrated HCl2. Predict how the fourth alcohol with the formula C4H10O would react if tested with:• 0.01 M KMnO4• concentrated HCl at room temperatureExtend FurtherUse your observations of the solutions formed in the previous experiments and yourunderstanding of alcohols to complete a table like the one shown below. Research the meltingand boiling points to verify your answers.arrow_forwardProvide the major organic product of the following reaction. + { A1C13arrow_forwardGive major and minor productsarrow_forward
- *GIVE EXPLANATIONS FOR ALL QUESTIONS (EXCEPT NOMENCLATURE). 9) Predict and draw the structure(s) of the product(s) of the following reactions. BRIEFLY JUSTIFY THE SPACING(S)THAT RESULT FROM THE REACTIONS WHERE APPLICABLE. OH Br NO₂ OH Cl₂ FeCl3 i CH3CH₂CCI AICI 3 HNO3 H₂SO4 AICI3 KMnO H₂Oarrow_forwardCompound A and compound B are in equilibrium. Write a stepwise mechanism from compound Ato compound B showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and products are formed. Show all lone pairs and formal charges. Lastly, explain which compound (Aor B) will be in higher concentration.arrow_forwardProvide the reagent(s) for these reactions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY