
(a)
Interpretation:
Condensed structural formula for acetyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
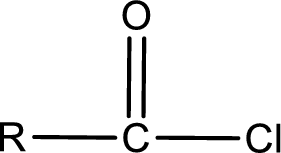
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent
(b)
Interpretation:
Condensed structural formula for 2-methylbutanoyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
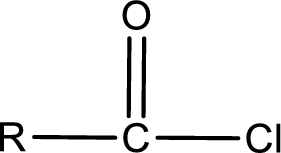
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent carboxylic acid from which the acid chloride is obtained. Name of the acid chloride is obtained by replacing “-oic acid” in the parent carboxylic acid name with “-oyl chloride”.
(c)
Interpretation:
Condensed structural formula for propionic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
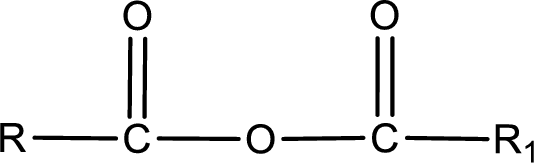
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an asymmetric one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
(d)
Interpretation:
Condensed structural formula for ethanoic methanoic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
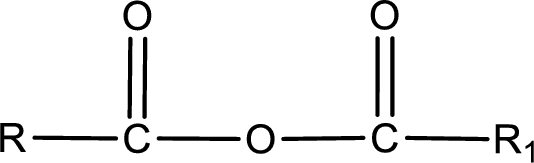
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an mixed one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
Trending nowThis is a popular solution!

Chapter 16 Solutions
General, Organic, and Biological Chemistry
- Which of the following compounds is a dihydroxy alcohol? a. 2-propanol b. 1,2-ethanediol c. 1,2,3-propanetriol d. ethanol e. both 1,2-ethanediol and 1,2,3-propanetriolarrow_forwardThe carboxylic acid compound is widely used as an antifungal agent a. oxalic b. benzoic c. salicylic d. aceticarrow_forwardWhat are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. benzoic acid and ethanol b. phenylic acid and ethanol c. ethanoic acid and benzene d. acetic acid and toluene e. phenylic acid and methanolarrow_forward
- 1. This structure will form when a hydroxyl group form a bond with a carbonyl group. a.Aldehyde b.Alcohol c.Hemiacetal d.straight chain formationarrow_forwardWhat kind of solvent ingredients is widely is usually used in mouthwash, perfumes and spray A. Ethly acetate B. Aliphatic alcohols C. Fragrant esters D. Phenols and phenol derivativesarrow_forward1. Chief organic component of vinegar a. acetic acid b. formic acid c. benzoic acid d. butanoic acid 2. This term means without water. a. carbonyl b. hydroxyl c. anhydride d. carboyl 3. Compounds containing the cyano group. a. nitriles b. amides c. amines d. nitrates 4. General formula of a Grignard reagent. a. RCOX b. RCN c. RCOOH d. RMgX 5. Organic derivatives of ammonia, derived from replacing one, two or all three hydrogens of the ammonia. a. amide b. amine c. cyan d. nitro 6. Sulfur analogs of alcohols where the O in R-OH is replaced by sulfur. a. Thioesters b. Thiols c. Thioaldehydes d. Thioethers 7. General formula of alkanes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 8. General formula of alkenes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 9. General formula of alkynes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 10. Which is soluble in water? a. methanol b. ethanol c. propanol d. all of the above 11. Which substance will have the highest boiling point? a. methanol b.…arrow_forward
- a. formaldehyde b. acetone c. benzaldehyde d. acetophenone which among the 4 samples contains the aldehyde group, which among the 4 samples does not contain the aldehyde group? Explainarrow_forwardEsters are formed by the reaction of carboxylic acids and acid in the presence of A. Alkanes B. Alcohols C. Alkenes D. Aldehydes E. Alkynesarrow_forwardAmide formation involves which two functional groups? A.carboxylic acid and alcohol B.alcohol and ketone C.carboxylic acid and amine D.amine and alcoholarrow_forward
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forwardA chemist wants to make an ester from conversion of alcohol or phenol, what can he used? a. benzamide b. acetic anhydride c. maleic anhydrate d. nonearrow_forwardWhich of the following molecules gives a condensation reaction with benzaldehyde? A. 4-methylbenzaldehyde B. 4-chlorobenzaldehyde C. 2,2-methyl propanoic acid D. 3-methylbutanoic acid E. 3-methylbenzaldehydearrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

