
Concept explainers
Interpretation:
To predict the structure of the coloured compound when the given colourless compound come in contact with the acid treated sheet.
Concept introduction:
Acid-base indicators are the compounds which changetheir colour on changing the pH. The reaction of the acid-base indicator can be represented by:
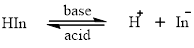
In organic compounds chromophoreis the unsaturated part that imparts colour. An auxochrome is a
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry (8th Edition)
- Suggest a possible structure for Compound X.arrow_forward(b) Give the products for the following chemical reactions. HO SOCI, CHO NACN (H) (D HCNarrow_forwardAn unknown has a chemical formula of C4H100. When chromic acid was added to the unknown, it turned green. When the Lucas reagent was added to a separate portion of the unknown, it turned cloudy in 5 minutes. When NaOH and Kl/iodine was added to the unknown, a yellow precipitate formed. What is the structure of the unknown compound?arrow_forward
- Excess ascorbic acid is excreted in the urine, the pH of which is normally in the range 4.8–8.4. What form of ascorbic acid would you expect to be present in urine of pH 8.4— free ascorbic acid or ascorbate anion? Explainarrow_forwardWhich Compound Is Oxidized To Benzoic Acid With K2Cr2O7 In Acidic Medium?arrow_forwardGive the product of the following reaction. HCIarrow_forward
- Compounds like amphetamine that contain nitrogen atoms are protonated by the HCl in the gastric juices of the stomach, and the resulting salt is then deprotonated in the basic environment of the intestines to regenerate the neutral form. Write proton transfer reactions for both of these processes.arrow_forwardMany multi-step organic reaction mechanisms involve proton transfer steps. For example, the first step of Fischer esterification of carboxylic acids(as shown with acedic acid) is activation of the acid by protonation. Based on your understanding of which reaction pathway is more favorable, explain why using chemical structures.arrow_forwardCalculate the percentage of free acid for (a) phenobarbital (it is an acid with pKa = 7.40) and (b) hexobarbital (also an acid with pKa = 8.4) at the physiological condition of pH 7.4.arrow_forward
- propanoic acid + methanol (in concentrated sulfuric acid)arrow_forwardA B с D AT&T 5G+ Lola wol o gº OEI . OEt + NaOH ethyl 4-aminobenzoale chloride salt NaOH + H₂O 3:37 PM + H₂O 0= OEt white solid علومة white solid white solid OEI 34% + + X . NaCl white sold NaCl HOE!arrow_forwardCaC2O4 + KMnO4 (in the presence of H2SO4)arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT





