
Concept explainers
(a)
Interpretation:
Among the given letters in the boxes which is appropriate for having
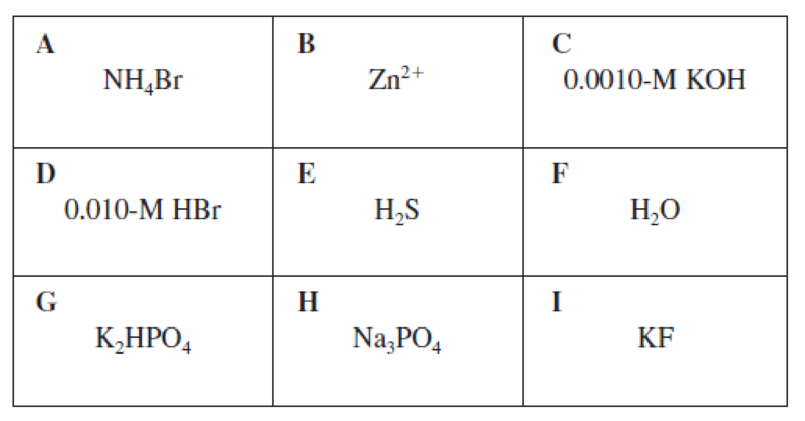
Concept Introduction:
(b)
Interpretation:
Among the given letters in the boxes which is appropriate for having a conjugate acid or conjugate base of another item in the grid has to be determined.
Concept Introduction:
Acid-base conjugate pairs differ by one
(c)
Interpretation:
Among the given letters in the boxes which is having a
Concept Introduction:
(d)
Interpretation:
Among the given letters in the boxes which is appropriately describing a polyprotic acid has to be determined.
Concept Introduction:
A polyprotic acid has more than one acidic hydrogen.
(e)
Interpretation:
Among the given letters in the boxes which is appropriate for describing
Concept Introduction:
If a compound can act both as an acid and as a base, the value of
(f)
Interpretation:
Among the given letters in the boxes which is appropriate for describing a basic salt has to be determined.
Concept Introduction:
A basic salt is a salt that contains an anion that is the conjugate acid of a weak acid.
(g)
Interpretation:
Among the given letters in the boxes which is appropriate for having
Concept Introduction:
(h)
Interpretation:
Among the given letters in the boxes which is appropriate for describing a Lewis acid has to be determined.
Concept Introduction:
Lewis acid is an electron pair acceptor.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Chemistry: The Molecular Science
- For oxyacids, how does acid strength depend on a. the strength of the bond to the acidic hydrogen atom? b. the electronegativity of the element bonded to the oxygen atom that bears the acidic hydrogen? c. the number of oxygen atoms? How does the strength of a conjugate base depend on these factors? What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide.arrow_forwardCalculate the pH of each of the following solutions. (a) 10.0 mL of 0.300 M hydrofluoric acid plus 30.0 mL of 0.100 M sodium hydroxide (b) 100.0 mL of 0.250 M ammonia plus 50.0 mL of 0.100 M hydrochloric acid (c) 25.0 mL of 0.200 M sulfuric acid plus 50.0 mL of 0.400 M sodium hydroxidearrow_forwardUsing the diagrams shown in Problem 10-37, which of the four acids is the weakest acid?arrow_forward
- Indicate whether each of the solutions in Problem 10-65 is acidic, basic, or neutral. a. 3.5 103 M b. 4.7 106 M c. 1.1 108 M d. 8.7 1010 Marrow_forwardMark each of the following statements True or False: a. The conjugate base of a strong acid is always a weak base. b. The conjugate acid of a strong base is always a weak acid. c. The stronger the acid, the weaker its conjugate base, and vice versa.arrow_forwardIn the following net ionic reaction, identify each species as either a Brnsted-Lowry acid or a Brnsted -Lowry base: CH3COO(aq)+HS(aq)CH3COOH(aq)+S2(aq). Identify the conjugate of each reactant and state whether it is a conjugate acid or a conjugate base.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





