
Concept explainers
(a)
Interpretation:
Hydrogen that can be exchanged with deuterium in a solution of
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form. Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomer is the location of hydrogen and double bond.

(b)
Interpretation:
Hydrogen that can be exchanged with deuterium in a solution of
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form. Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomer is the location of hydrogen and double bond.

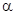 -carbon is the carbon adjacent to a carbonyl carbon.
-carbon is the carbon adjacent to a carbonyl carbon.
(c)
Interpretation:
Hydrogen that can be exchanged with deuterium in a solution of
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form. Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomers is the location of hydrogen and double bond.

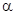 -carbon is the carbon adjacent to a carbonyl carbon.
-carbon is the carbon adjacent to a carbonyl carbon.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Essential Organic Chemistry (3rd Edition)
- Please draw the structure of the possible product for the following reaction, and how does the oxidation state of Rh formally change in this reaction? 17. (a) trans-Ir(CO)Cl(PPH3)2 + H2 → (b) H;C-Mn(CO); + P(CH;)Ph2 → (no gas evolved)arrow_forwardWhat two components are needed to prepare para red by azo coupling?arrow_forwardWhich group elements show maximum catalytic activity for hydrogenationreactions?arrow_forward
- Which of the following compounds can be prepared by radical halogenation with minimum complication due to the formation o isomeric by-products? CI Br Br Br CIarrow_forwardCompare and contrast the physical and chemical properties of: • propan-1-ol and propan-2-ol; • (E)-butenedioic acid (fumaric acid) and (Z)-butenedoic acid (maleic acid)arrow_forwardWhat changes in color take place when KMnO4 reacts with an alkene?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





