
A small oven consists of a cubical box of dimension
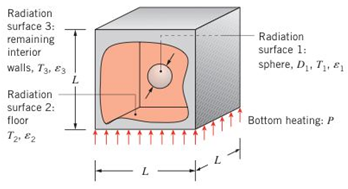
(a) Find the following view factors: F12, F13, F21, F31, F23, F32, F33.
(b) Determine the temperature of the floor and the net rate of heat transfer leaving the sphere due to radiation. Is the sphere under steady-state conditions?
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Fundamentals of Heat and Mass Transfer
- Question 5: Z=62 a. An iron sphere of mass (Z + 300)g is kept in a container having boiling water (100 °C). If the temperature of the sphere is 25.5°C, how much heat energy is absorbed by the iron sphere? Consider the specific heat of iron as 452J/kg. b. The wall of an industrial furnace is constructed from (Z + 3) cm thick fireclay brick having a thermal conductivity of 1.7 W/mK. Measurements made during steady-state operation reveal temperatures of 530°C and 375°C at the inner and outer surfaces, respectively. Find the rate of heat loss through a wall which is (Z + 5) cm by (Z + 3) m on a side.arrow_forwardThe diagram below illustrates a simplified version of the Jet Propulsion Laboratory's 25-ft space simulator chamber. Within it, tests are run for deep space probes and their components. In this particular experiment, a sensor assembly with its housing is placed inside. The sensor-housing electronics create heat such that q=3181.0 W exits the assembly's top surface into the chamber. The chamber itself is under vacuum, and its walls are at cryogenic temperatures to simulate conditions in space. Surfaces 1 and 2 are flat. Assume they are flush. The top surface of the chamber, Surface 4, can be approximated as a perfectly insulated, flat ceiling. The exposed surface of the floor, Surface 2, is a black, donut- shaped surface. Additional surface information can be seen below, including various parameters and dimensions. Assume all surfaces are opaque and diffuse, and conditions are at steady-state. PARAMETERS & DIMENSIONS Surface 1: A = 7.07 m²; diameter d; = 3 m; &₁ = 0.5; Surface 2: A₂ =…arrow_forwardA box made of copper plate with outside dimensions of 5 ft by 5 ft by 6-in.is suspended horizontally in a large room where the air and wall temperaturesare 80°F. The box contains a heating coil which maintains a temperature of500 °F on all external surfaces. What is the total heat loss from the box inBtu/hr.arrow_forward
- A machine element as seen in the figure is made of pure copper. It has an inner diameter of D;=2.9 cm, outer diameter of Do=5.9 cm, and height of H=19 cm. The initial temperature of the element is 749 K, and then suddenly placed in an environment which has a temperature of Too=300O K. The temperature of the element is measured as 489 K, 4.8 minutes after the cooling process. If the lumped system method is applicable, determine the heat transfer coefficient of the environment, in W/(m²K). Note: All the surfaces should be considered to calculate the surface area. Not: Yüzey alanı hesaplamak için tüm yüzeyler dikkate alınmalıdır. Properties of pure copper: k=370 W/(mK), C=920 J/(kg°C), p=8933 kg/m³ H Dịarrow_forwardThe hollow sphere has an outside radius of 1 m and is made of polystyrene foam with a thickness of 1 cm. A heat source inside keeps the inner surface 5.20°C hotter than the outside surface. How much power is produced by the heat source? The thermal conductivity of polystyrene is 0.033 W/m°C.arrow_forwardThank you for answering this question. Please you specify each step as soon as possible. Good Luck! Thank you. Two plane coaxial disks are seperated by a distance L = 0,30 m. The lower disk (A1) is solid with a diameter Do = 0,6 m and a temperature T1 = 300 K. The upper disk (A2) has the outer diameter Do = 0,3 m and a temperature T2 = 1000 K. Assuming the disks to be blackbodies, calculate the net radiative heat exchange from upper to lower disk.arrow_forward
- 4. Two aluminum blocks with different dimensions, but the same mass are shown in the figure. The dimensions of block 1 are 0.25 cm in thickness, 3 cm in width, and 36 cm in length. The dimensions of block 2 are 3 cm for each side of the cube. Aluminum has a density of 2700 kg/m³ and a specific heat capacity of 900 J/kg-°C. The value of the heat transfer coefficient to the surrounding air is 10 W/m²-°C. 0.25 cm 36 cm 3 cm 3 cm Block 1 3 cm Block 2 3 cm Complete the following. (a) Calculate thermal time constant (in min) for both blocks. (b) Each block is heated to an initial temperature 120 °C and then allowed to cool in the surrounding air which is at 20 °C. Write a script file in MATLAB® that plots the temperature as a function of time, T(t), for both blocks on the same graph. Plot the temperature of block 1 using a solid black line and the temperature of block 2 using a dotted black line. Plot for a duration equal to four of the longer time constant between blocks 1 and 2. The time…arrow_forwardA radiator plate of 0.2-m (wide) x 0.4-m (height) is in a room of 22°C. One side (the hot side facing the room) of the radiator plate is maintained at a temperature of 80°C, the other sides are assumed to be insulated, as shown in Figure 1. If the following assumptions can be made: 1) steady operating conditions exist; 2) the air an ideal gas; 3) the local' atmospheric pressure is 1.00 atm: (a) Briefly and qualitatively analyze the four different locations for the radiators marked A), B), C) and D), as shown in Figure 1, and rank them in terms of heat transfer efficiency by natural convection. (b) Determine the rate of heat transfer from the radiator plate by natural convection if the plate is A) vertical and C) horizontal with the hot surface facing down. Compare the two and provide a brief discussion of the findings. wall A 0.4 mi C 0.4 m 0.4 m B floor ceiling 0.4 m wall Nu Nu=0.59Ra Nu-0.1 Raa 0.387Ral Nu- - {0.825 + 11 + (0.492/Pr)162) (complex but more accurate) Use vertical plate…arrow_forwardCalculate the heat transferred from the cube-shaped iron mass (a = 30 cm) to the environment at 20 ° C with all surfaces at a temperature of 100 ° C. (Assume that the heat is only from the surfaces, the inner parts of the surfaces are insulated.)arrow_forward
- On a mild Saturday morning while people are working inside, the furnace keeps the temperature inside the building at 23°C. At noon the furnace is turned off, and the people go home. The temperature outside is a constant 14°C for the rest of the afternoon. If the time constant for the building is 3 hr, when will the temperature inside the building reach 19°C? If some windows are left open and the time constant drops to 2 hr, when will the temperature inside reach 19°C? If the time constant for the building is 3 hr, the temperature inside the building will reach 19°C about hr after noon. (Round to the nearest tenth as needed.) If the time constant for the building drops to 2 hr, the temperature inside the building will reach 19°C about (Round to the nearest tenth as needed.) hr after noon. Carrow_forwardSipho’s mother is making a fire in the stove inside a shack using coal. A fan is used to extract the flue gasfrom the fire via a chimney made of zinc material. The wall thickness of Chimney is L=4mm and the walltemperature of the chimney on the flue gas side is T_si=60˚C. Sipho, playing outside next to the chimneyexperiences an ambient temperature of 20˚C. The surrounding temperature is T_sur=35˚C and the emissivityis ε=0.95. Assume the convection coefficient is 20W/m2K and the chimney is grey material.Under these conditions determine the outer surface (air side) temperature T_so=? of the chimney.arrow_forwardA coil-shaped cooling pipe is made of SS-304 material. This pipe is 1 ft long, 0.4 inch outside diameter, and inch inside diameter. This coil cooling pipe is used to cool the water in the bath. The temperature of the inner coil pipe is 40oF while the outer coil in contact with water is 80oF. The thermal conductivity of SS-304 is a function of temperature where k(T) = 7.75 + (7.78 x 10-3).T where k is in Btu/h.ft.oF and T is in oF. Calculate the rate of heat dissipation in watts! (1287.7)arrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
