
What products would result from the following processes?
Write an equation for each reaction.
a.
b.
c.
d.
e.
(a)
Interpretation:
The product formed when
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
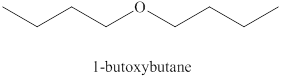
Alcohols on heating at
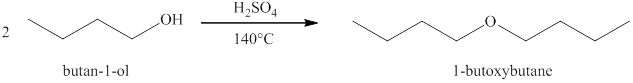
Figure 1
The product formed when
(b)
Interpretation:
The product formed by the excess oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed by the excess oxidation of
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
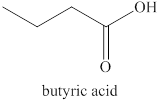
The primary alcohol,

Figure 2
The product formed by the excess oxidation of
(c)
Interpretation:
The product formed by the controlled oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
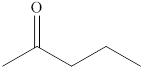
The compound
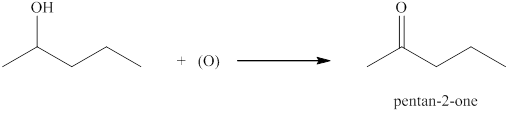
Figure 3
The product formed when
(d)
Interpretation:
The product formed when
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
![]()
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Alcohols on heating at temperature
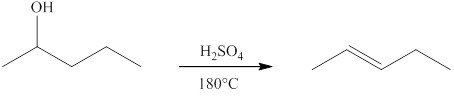
Figure 4
Interpretation:
The product formed when
(e)
Interpretation:
The product formed by the controlled oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
No product is formed by the controlled oxidation of
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
The controlled oxidation of
No product is formed by the controlled oxidation of
Want to see more full solutions like this?
Chapter 13 Solutions
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
- 1. Which of the following statements is true? I. Aldehydes and ketones both contain a hydroxyl group. II. The names for aldehydes and ketones are derived from the name of the longest carbon chain that contains the carbonyl group. III. The aldehyde and ketone with a molecular formula of C3H6O are constitutional isomers. IV. 2-Propanone is immiscible in water. A. I & II B. II & II C. I & III D. I & IV 2. Whicb of the following is the correct IUPAC name of the structure below?arrow_forwardWhich of the following is not a physical property of alcohols or phenols? Select one: A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Phenols are generally only slightly soluble in water. C. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes. D. The hydroxyl group of an alcohol is nonpolar.arrow_forwardWhich of the following statements is true? a. Both aldehydes and ketones are easily oxidized.b. Neither aldehydes nor ketones are easily oxidized.c. Aldehydes are easily oxidized, but ketones are not.d. Ketones are easily oxidized, but aldehydes are not. 2. Which of the following is obtained by oxidizing hexanal? a. hexanol b. 2-hexanone c. hexanoic acid d. none of these 3. Which of the following oxidizing agents oxidizes aldehydes in the body? a. NADH b. NAD+c. O2 d. none of these 4. Which of the following will be obtained by the reaction of pentanal with H2 in the presence of a transition metal catalyst? a. pentene b. 1-pentanolc. 2-pentanol d. pentanoic acid 5. The reaction of which combination of reactants result in the formation of a hemiacetal? a. ketone and alcohol b. ketone and aldehydec. ketone and carboxylic acid d. none of thesearrow_forward
- 1. When an aldehyde is reduced with hydrogen (H2) gas in the presence of a transition metal catalyst, what type of product is formed? A. primary alcohol B. secondary alcohol C. tertiary alcohol D. carboxylic acidarrow_forwardPlease explain the chosen letter. Which is NOT a physical property of alcohols or phenols? a. Phenols are generally only slightly soluble in water. b. The solubilities of normal primary alcohols in water decrease with increasing molecular weight. c. The hydroxyl group of an alcohol is nonpolar. d. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes.arrow_forwardWhich of the following compounds can react with C2H5MgBr to give 3-pentanol? a.acetone b.ethanal c.acetic acid d.ethyl formatearrow_forward
- 1. What is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.2. How do you neutralize the acetic acid regenerated in the reaction? Write the balanced chemical reaction.arrow_forwardWhat is the name of the major product formed during the reaction between berzoyl chloride and phenol? a. phenyl benzoate b. benzyl ester C. cyclopentanoate d. benzyl phenoate e. benzenecarboxylic acid O a O b O c earrow_forwardWhich of the following compounds is a dihydroxy alcohol? a. 2-propanol b. 1,2-ethanediol c. 1,2,3-propanetriol d. ethanol e. both 1,2-ethanediol and 1,2,3-propanetriolarrow_forward
- Consider the structure of cyclohexene, if it undergoes epoxidation followed by exposure to water, which of the following final product is formed? a. Cyclohexan-1,2,-diol b. Cyclohexane c. Cyclohexanone d. Hexan-1,-6-dioic acidarrow_forward3. Write the following reaction including structures of the reactants and products a. Oxidation of 1-propanol b. Oxidation of 2-pentanol c. Oxidation of 3-methyl-1-cyclohexanolarrow_forward1.Give the common name of 2,2-dimethyl-1-propanethiol and give the complete equation as the compound is exposed to oxidation. 2.What will be the product of the compound in #1 if added with NaOH?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





