
Concept explainers
12-27 Explain why each name is incorrect and then write a correct name.
- 2-Ethyl-l-propene
- 5-lsopropylcyclohexene
- 4-Methyl-4-hexene
- 2-sec-Butyl-l-butene
- 6,6-Dimethylcyclohexene
- 2-Ethyl-2-hexene
(a)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P
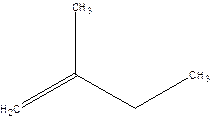
2-Methyl-but-1-ene.
Explanation of Solution
2-Ethyl-1-propene.
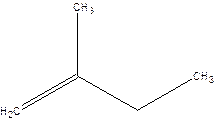
Error- longest chain not correctly located.
Correct name-
2-Methyl-but-1-ene.
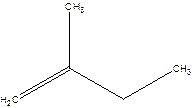
(b)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P
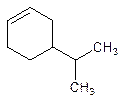
4-Isopropylcyclohexene.
Explanation of Solution
5-Isopropylcyclohexene.
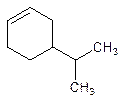
Error- position of the substituent not correctly mentioned.
Correct name-
4-Isopropylcyclohexene.
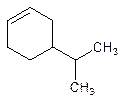
(c)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P

3-Methyl-2-hexene.
Explanation of Solution
4-Methyl-4-hexene.
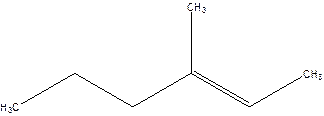
Error- position of the double bond not correctly mentioned.
Correct name-
3-Methyl-2-hexene.
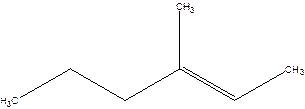
(d)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P
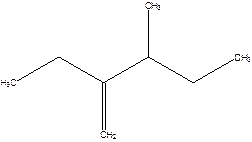
2-Ethyl-3-methyl -1-pentene.
Explanation of Solution
2-sec-butyl-1-butene.
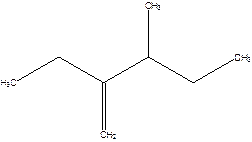
Error- longest chain not correctly determined.
Correct name-
2-Ethyl-3-methyl -1-pentene.
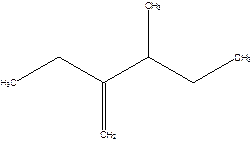
(e)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P
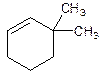
3,3-Dimethylcyclohexene.
Explanation of Solution
6,6-Dimethylcyclohexene.
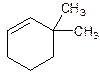
Error- position of substituents not correctly determined.
Correct name-
3,3-Dimethylcyclohexene.
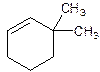
(f)
Interpretation:
To identify the error in name of the given compound and write the correct IUPAC names.
Concept Introduction:
The rules to write the IUPAC names are as follows-
- The longest carbon chain is identified, and the root name is given accordingly.
- All the substituents attached to the root chain are determined.
- The position of the substituents is so assigned that the sum of their positions comes to be the least of all possible positions.
- The prefix di, tri, tetra is used if the same substituent is present two, three and four times respectively.
- All the substituents in the name are written in alphabetical order.
- For a cyclic hydrocarbon the prefix cyclo- is used.
Answer to Problem 17P

3-Methyl-3-Heptene.
Explanation of Solution
2-Ethyl-2-hexene.

Error- longest chain not correctly determined.
Correct name-
3-Methyl-3-Heptene.

Want to see more full solutions like this?
Chapter 12 Solutions
Introduction To General, Organic, And Biochemistry
- 12-23 Draw a structural formula for each compound. 3-Chloropropene 3-Methylcyclohexene 1,2-Dimethylcyclohexene /runs-3,4-Dimethyl-3-heptene Cydopropene 3-Hexynearrow_forward13-53 Write the structural formula for the product of each reaction.arrow_forwardWhat is the IUPAC name of the following compound? CH3CH2CH2-C°C-CH(CH3)2 O 1-isopropyl-1-pentyne O 2-methyl-3-heptyne O 6-methyl-4-heptyne O isopropyl propyl ethynearrow_forward
- Draw the structure of the given compounds. Identify if the given name is correct or incorrect. Write the word ACCURATE if it is correct. If not, then provide the exact IUPAC Name. 1-ETHYLCYCLOPENT-2-ENE 7-HYDROXYNON-3-ENE 4-ETHYL-5,5-DIMETHYLNON-8-EN-1-YNE 7-BUTYNYLDODEC-1-ENE 6-PROPYLCYCOHEPTENEarrow_forwardWhat would be the IUPAC name of the following What would be the IUPAC name of the following structure? structure? 5-isopropyloct-4-ene-2-yne 4-ethyl-7-propyldec-2-ene 6-methyl-5-propylhept-4-ene-2-yne 7-ethyl-4-propyldec-1-ene 2-methyl-3-propylhept-3-ene-5-yne 4-ethyl-7-propyldec-8-ene 4-isopropyloct-4-ene-6-yne 7-ethyl-4-propyldec-2-ene Which of the following structures would correspond to 1-fluoro-2,2,5,5-tetramethylhexane? What would be the IUPAC name of the following structure? Br А. с. F. 'F D. 2-bromohexane 5-bromohexane O B 5-bromo-5-methylpentane 1-bromo-1-methylpentane O A Photo Grid B.arrow_forwardWhat is the parent chain for the following compound? O Heptane O Octane Nonane O Decanearrow_forward
- What is the correct IUPAC name for the following compound? 3-propylhept-2-yne O 1-cyclopropyl-1-hexyne O 1-cyclopropane-4-nonyne O 6-cyclopropyl-5-hexynearrow_forwardWhat is the IUPAC name for the following compound? O butylcyclohexane O 1-cyclohexylbutane 1-cyclohexylpropane O sec-butylcyclohexane O ethylcyclohexane O isopropylcyclohexane O cyclohexylethane O t-butylcyclohexane isobutylcyclohexane O propylcyclohexanearrow_forward12-40 Define alkene addition reaction. Write an equation for an addition reaction of propene.arrow_forward
- Name and draw structural formulas for all pos sible monochlorination products that might be formed in each reaction.arrow_forward12-58 Show how to convert ethylene to these compounds (a) Ethane(b) Ethanol (c) Bromoethane(d) 1,2-Dibromoethane (e) Chloroethanearrow_forward12-29 Which of these alkenes show cis-trans isomerism? For each that does, draw structural formulas for both isomers. (a) 1-Hexene(b)2-Hexene (c) 3-Hexene(d)2-Methyl-2-hexene (e) 3-Methyl-2-hexene(f)2,3-Dimethyl-2-hexenearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




