
Concept explainers
Interpretation:
The structural formulas and the IUPAC names for the
Concept introduction:
The systematic naming of organic compound is given by
Rules for writing IUPAC name from structural formula are:
• First identify the longest carbon chain.
• The next step is to identify the groups attached to the longest chain.
• Identify the position, location, and number of the substituents bonded to the carbon chain.
• Use prefix di, tri, tetra if same type of substituents are present.
• Name the substituents in alphabetical order.
Answer to Problem 12.9E
The structural formulas and the IUPAC names of the
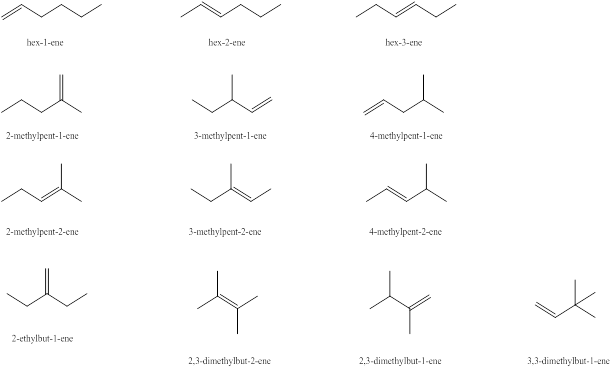
Explanation of Solution
The first alkene isomer of
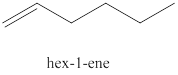
Figure 1
The second alkene isomer of
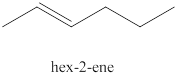
Figure 2
The third alkene isomer of
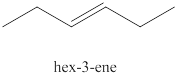
Figure 3
The fourth alkene isomer of
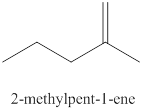
Figure 4
The fifth alkene isomer of
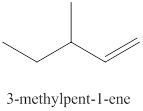
Figure 5
The sixth alkene isomer of
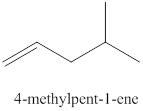
Figure 6
The seventh alkene isomer of
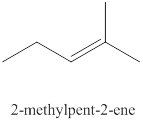
Figure 7
The eighth alkene isomer of
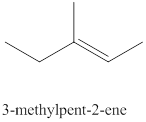
Figure 8
The ninth alkene isomer of
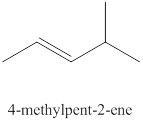
Figure 9
The tenth alkene isomer of
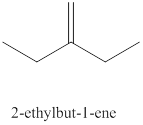
Figure 10
The eleventh alkene isomer of
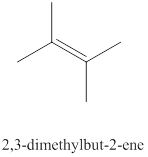
Figure 11
The twelfth alkene isomer of
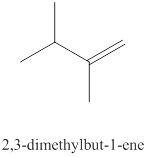
Figure 12
The thirteenth alkene isomer of
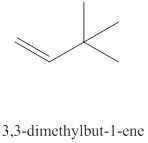
Figure 13
The structural formulas and the IUPAC names of the
Want to see more full solutions like this?
Chapter 12 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
- What is the difference in bonding and in general molecular formula between an alkene and a cycloalkane with the same number of carbon atoms?arrow_forwardWhat is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forwardWrite the molecular formula of each alkane.arrow_forward
- Gasohol is a mixture of 90% gasoline and 10% ethanol, CH 3CH 2OH. Ethanol is considered an environmentally friendly fuel additive because it can be made from a renewable source—sugarcane. Ethanol burns in air to form CO 2 and H 2O, and, like the combustion of alkanes, this reaction also releases a great deal of energy. Write a balanced equation for the combustion of ethanol.arrow_forwardWhat is the molecular formula for a monocyclic hydrocarbon with 14 carbons and 2 triple bonds?arrow_forwardWrite condensed structural formulas and give the IUPAC name for each structural isomer of C2H4Cl2arrow_forward
- In organic chemistry, is it only when naming cycloalkanes that you do not put a hyphen between the substituent and the parent ring?arrow_forwardExplain geometric isomerism in alkenes. How do the properties of geometric isomers differ from one another?arrow_forwardDraw the line structures and give the names of the simplest straight chain alkenes (the double bond between C1 – C2) containing seven to twelve carbons.arrow_forward
- 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used as a standard in determining the octane rating of gasoline a. Draw the structural formulas for at least ten chain isomers of octane. b. Give the IUPAC name of each. C. Which of the isomers that you have drawn has the highest boiling point? Which has the lowest boiling point? Rationalize. 2. Which of the following structural formulas represent identical compounds and which represent constitutional/structural isomers? Identical compounds: Constitutional isomers: a). CH3CH2CHCH3 e). CH2CH2CHCH3 CH3 i). CH3-C-CI ČI CI CI CH3 CH2CI b). CH3-C-CH3 f). CH3CH2CH2CH,CI j). CICH2 CI CH3 g). CICH,CHCH3 CH2CI k). CH3-CH-CH3 CI c). CH,CHCHCH3 CI h). CH3CHCH2CH2CI CH2CH3 1). CH3CHCI d). CI CIarrow_forwardDefine Natural Occurrence of Alkanes ?arrow_forwardWrite structures of different isomers formed by C6 H10 . Also write IUPAC names of the all the isomersarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



