
Concept explainers
(a)
Interpretation:
The model structure has to be converted into line drawing along with identification of Functional Group.
Concept Introduction:
Line structure is a simple and quick way to represent organic molecules without showing carbons and hydrogens present. Drawing a molecule in this way is very simple:
It follows the following guide line:
- Each carbon- carbon bond is represented by a line.
- Anywhere a line ends or begins, as well as any vertex where two lines meet represents a carbon atom.
- Any atom other than another’s carbon or hydrogen attached to a carbon must be shown.
- Since a neutral carbon atom form four bonds, all bonds not shown for any carbon are understood to be the number of carbon – hydrogen bonds needed to have.
Functional group: Organic compounds can be classified into various families according to the

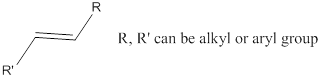
Ether: A class of organic compounds, where oxygen is attached to two alkyl or aryl groups.
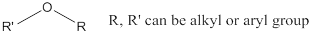
(b)
Interpretation:
The model structure has to be converted into line drawing along with identification of Functional Group.
Concept Introduction:
Line structure is a simple and quick way to represent organic molecules without showing carbons and hydrogens present. Drawing a molecule in this way is very simple:
It follows the following guide line:
- Each carbon- carbon bond is represented by a line.
- Anywhere a line ends or begins, as well as any vertex where two lines meet represents a carbon atom.
- Any atom other than another’s carbon or hydrogen attached to a carbon must be shown.
- Since a neutral carbon atom form four bonds, all bonds not shown for any carbon are understood to be the number of carbon – hydrogen bonds needed to have.
Functional group: Organic compounds can be classified into various families according to the functional groups they contain a functional group is a part of a larger molecule and is composed of a group of atoms that has characteristic structure and chemical reactivity.
Functional group:
An atom or group of atoms which has physical and chemical properties is known as functional group.
Ketone: A compound containing a carbonyl group

Alkene: Unsaturated hydrocarbons containing double bonds.
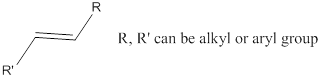
Ether: A class of organic compounds, where oxygen is attached to two alkyl or aryl groups.
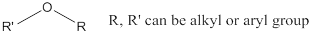
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Potentiometric titration curve is given below, which is obtained during the potentiometric titration between strong base KOH (0.2 M) with strong acid HI, label the point in the curve from the following options. If more than one points are present than write as x, y(means separate by using comma) a)The point where pH is because of excess OH - ions. b) The point where pH is only because of HI in water. c)The point where [HI]= [I] in water. d)The point where pH=pka e) The point where all HI is neutralized. f) The point where pH corresponds to solution of [I- ] in water. 14 13 12 11 10 9 pH 6. TITIT TITarrow_forwardUsing the picture provided, match the correct name of each of the functional groups highlighted in blue.arrow_forwardIdentify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forward
- Draw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forwardWhen Alfred Werner was developing the field of coordination chemistry, it was argued by some that the optical activity he observed in the chiral complexes he had prepared was due to the presence of carbon atoms in themolecule. To disprove this argument, Werner synthesized a chiral complex of cobalt that had no carbon atoms in it, and he was able to resolve it into its enantiomers. Design a cobalt(III) complex that would be chiral if it could be synthesized and that contains no carbon atoms.arrow_forwardGive at least 5 examples of biological compounds having an alcohol functional group and identify the biochemical importance of each compound.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





