
Concept explainers
(a)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
The structural formula of the given compound is shown below as,
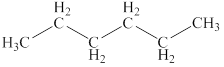
Explanation of Solution
The given compound is
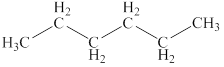
Figure 1
The structural formula of the given compound is shown in Figure 1.
(b)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
The structural formulas of the given compound are shown below as,
Explanation of Solution
The given compound is

Figure 2
The structural formulas of the given compound are shown in Figure 2.
(c)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
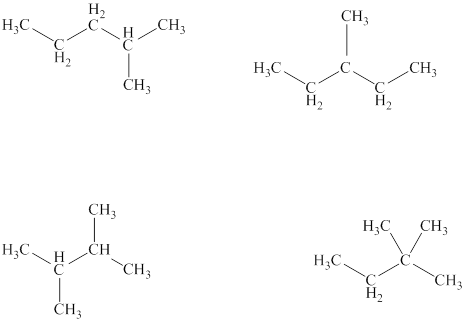
The structural formulas of the given compound are shown below as,
Explanation of Solution
Conformers are the type of isomers that can be interconverted into one other by simple rotation of bond.
The given compound is

Figure 3
The structural formulas of the given compound are shown in Figure 3.
(d)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
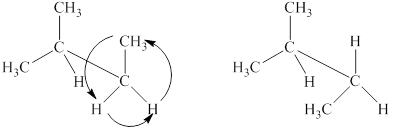
The structural formulas of the given compound are shown below as,
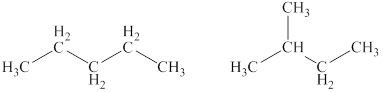
Explanation of Solution
The given compound,
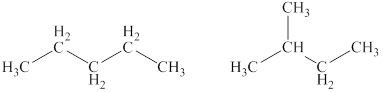
Figure 4
The structural formulas of the given compound are shown in Figure 4.
(e)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
The structural formula of the given compound is shown below as,

Explanation of Solution
The given compound is

Figure 5
The structural formula of the given compound is shown in Figure 5.
(f)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
The structural formulas of the given compound are shown below as,
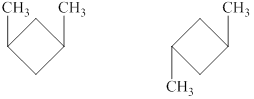
Explanation of Solution
The given compound,

Figure 6
The structural formulas of the given compound are shown in Figure 6.
(g)
Interpretation:
The structural formula of the given compound is to be predicted.
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called as isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 11.55E
The structural formula of the given compound is shown below as,
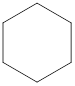
Explanation of Solution
The given compound,

Figure 7
The structural formula of the given compound is shown in Figure 7.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry for Today: General Organic and Biochemistry
- Is the general formula of a cycloalkanes the same as the general formula of an alkane, CnH2n+2? Draw any structural diagram to illustrate your answer.arrow_forwardHow does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardDistinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





