
Concept explainers
(a)
Interpretation: Structural formulae for the alkene that gives the indicated product should be drawn.
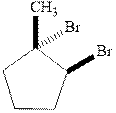
Concept introduction:
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
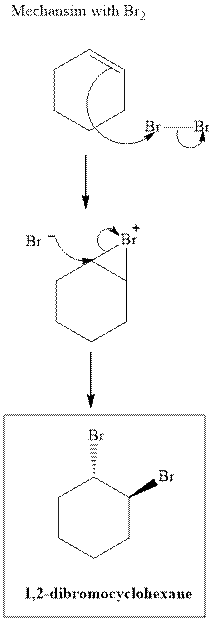
(b)
Interpretation: Structural formulae for the alkene that gives the indicated product should be drawn.
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
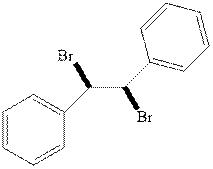
(c)
Interpretation: Structural formulae for the alkene that gives the indicated product should be drawn.
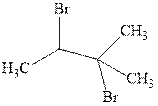
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- When propene reacts with gaseous hydrogen bromide, HBr, two products, 1-bromopropane and 2-bromopropane are formed. The reaction is a two-step process in which the electrophilic attack occurs in the first step. Identify the electrophile in this reaction Draw a diagram showing the first step of the reaction that leads to the production of 2-bromopropane.arrow_forwardIdentify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardSynthesize the product from the given material. Give the reagents necessary and draw out any intermediate products along the way.arrow_forward
- 1,3-Cyclohexadiene and 1,4-cyclohexadiene can be distinguished by reaction with ozone and then treatment with zinc. Draw the structures of the products produced by ozonolysis of the two compounds.arrow_forwardWhat are the various ways by which alkenes may be synthesized?arrow_forwardDraw the products expected from the addition of bromine to an alkene and to an alkyne.arrow_forward
- Name three alkenes that yield 3-methylpentane on catalytic hydrogenation.arrow_forwardWrite the structure of the major products in the following reactions and name them.arrow_forwardDraw the structure of each product from the reaction of benzene with 2-chloro-1-methylcyclohexane using AlCl 3 as the catalyst and Identify the major product.arrow_forward
- By taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forwardDraw the products (including stereoisomers) formed when 2- methylhex-2-ene is treated with HBr in the presence of peroxides.arrow_forwardAcid-catalyzed dehydration of 3-methyl-2-pentanol gives three alkenes: 3-methyl-1-pentene, 3-methyl-2-pentene, and 3-methylenepentane. Draw the structure of the carbocation intermediate leading to the formation of 3-methyl-2-pentene.arrow_forward
