
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 1, Problem 53P
(a) Consider a carbon atom in its ground state. Would such an atom offer a satisfactory model for the carbon of methane? If not, why not? (Hint: Consider whether a ground state carbon atom could be tetravalent, and consider the bond angles that would result if it were to combine with hydrogen atoms.)
(b) Consider a carbon atom in the excited state:
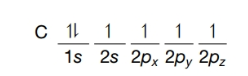
Excited state of a carbon atom
Would such an atom offer a satisfactory model for the carbon of methane? If not, why not?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(ii) What are the two parts of a wavefunction?
(iii) What is the importance of squaring a wavefunction?
(iv) Where is a wavefunction obtained from?
QUESTION 4
Draw the best Lewis structures for the substances below. Show your thinking and method.
(a)
(b)
Oxalic acid, H2C2O4
Bromate ion, BrO3-
XEOF2
CH3CN
QUESTION 5
Methanol (CH3OH) is a liquid at room temperature with a density of 7.91 ×102 kg/m³.
In a certain experiment, from the reaction of 2.91 mL of methanol with 2.88 g of oxygen, 2.27 g
of carbon dioxide was obtained.
(a)
(b)
What type(s) of chemical reaction is (are) occurring in this experiment?
What equipment do you think was used to measure the volume of methanol in this
experiment?
Calculate the percent yield of the carbon dioxide in this experiment.
(c)
5
(1)Draw a ground state energy level diagram for
a silicon atom that shows where all of the
electrons would be (n, L, ml and show spin up
or spin down with an arrow). (2) Determine the
values of L and S for the ground state using
Hund's rule. (3) For chlorine, repeat steps 1 and
2.
Q2.
22
(a)
Write down the Hamiltonian of a molecule having M nuclei and N electrons with a dear definition for each term.
(b) Derive the binding energy for the H2 molecule in terms of Coulomb (J), exchange (K) and overlap (S) integrals
using valence bond (VB) theory. Write down the singlet and triplet wavefunctions for an H₂ molecule using VB
theory.
(c) Draw the MO-diagram for a general diatomic molecule. Show the electronic configuration of F2*, F2 and discuss
their bond properties and relative stabilities.
Chapter 1 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. 1 - Prob. 1PPCh. 1 - Prob. 2PPCh. 1 - Prob. 3PPCh. 1 - Prob. 4PPCh. 1 - Prob. 5PPCh. 1 - Prob. 6PPCh. 1 - Prob. 7PPCh. 1 - Prob. 8PPCh. 1 - Prob. 9PPCh. 1 - Prob. 10PP
Ch. 1 - Prob. 11PPCh. 1 - Prob. 12PPCh. 1 - Prob. 13PPCh. 1 - Prob. 14PPCh. 1 - Prob. 15PPCh. 1 - Prob. 16PPCh. 1 - Prob. 17PPCh. 1 - Prob. 18PPCh. 1 - Prob. 19PPCh. 1 - Prob. 20PPCh. 1 - Prob. 21PPCh. 1 - Practice Problem 1.22 Which of the following...Ch. 1 - Prob. 23PPCh. 1 - Prob. 24PPCh. 1 - Practice Problem 1.25
What do the bond angles of...Ch. 1 - Prob. 26PPCh. 1 - Practice Problem 1.27
Use VSEPR theory to predict...Ch. 1 - Practice Problem 1.28 Predict the bond angles of...Ch. 1 - 1.29 Which of the following ions possess the...Ch. 1 - 1.30 Write a Lewis structure for each of the...Ch. 1 - Prob. 31PCh. 1 - Add any unshared electrons to give each element an...Ch. 1 - Prob. 33PCh. 1 - What is the molecular formula for each of the...Ch. 1 - Prob. 35PCh. 1 - Prob. 36PCh. 1 - 1.37 Write bond-line formulas for all of the...Ch. 1 - Prob. 38PCh. 1 - Prob. 39PCh. 1 - Prob. 40PCh. 1 - Prob. 41PCh. 1 - (a) Cyanic acid (HOCN) and isocyanic acid (HN=C=O)...Ch. 1 - Consider a chemical species (either a molecule or...Ch. 1 - 1.44 Consider a chemical species like the one in...Ch. 1 - 1.45 Consider another chemical species like the...Ch. 1 - Draw a three-dimensional orbital representation...Ch. 1 - Ozone (O3) is found in the upper atmosphere where...Ch. 1 - Write resonance structures for the azide ion, N3....Ch. 1 - Write structural formulas of the type indicated:...Ch. 1 - Prob. 50PCh. 1 - 1.51 In Chapter 15 we shall learn how the...Ch. 1 - Prob. 52PCh. 1 - (a) Consider a carbon atom in its ground state....Ch. 1 - Open computer molecular models for dimethyl ether,...Ch. 1 - Boron is a group IIIA element. Open the molecular...Ch. 1 - 1.56 There are two contributing resonance...Ch. 1 - Prob. 1LGPCh. 1 - Consider the compound with the following condensed...Ch. 1 - Consider the compound with the following condensed...Ch. 1 - Consider the compound with the following condensed...Ch. 1 - Consider the compound with the following condensed...Ch. 1 - Consider the compound with the following condensed...Ch. 1 - Prob. 7LGPCh. 1 - Prob. 8LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
If isomer A is heated to about 100 C, a mixture of isomers A and B is formed. Explain why there is no trace of ...
Organic Chemistry
45. Consider the reaction:
A reaction mixture in a 3.67 L flask at a certain temperature initially con...
Chemistry: Structure and Properties
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Q1. A chemical system produces 155 kJ of heat and does 22 kJ of work. What is ΔE for the surroundings?
a) 177 k...
Chemistry: A Molecular Approach (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Schrodinger and de Broglie suggested a ‘Wave—particle duality" for small particles—that is, if electromagnetic radiation showed some particle-like properties, then perhaps small punicles might exhibit same wave-like properties. Explain. How does the wave mechanical picture of the atom fundamentally differ from the Bohr model? How do wave mechanical arbitals differ from Bohr’s orbits? What does it mean to say that an orbital represents a probability map for an electron?arrow_forwardDo atoms in excited states emit radiation randomly, at any wavelength? Why? What does it mean to say that the hydrogen atom has only certain discrete energy levels available? How do we know this? Why was the quantization of energy levels surprising to scientists when it was first discovered?arrow_forwardWhat are the General Features of the Hammond Postulate ?arrow_forward
- Three of the term symbols that correspond to a nd3 electron configuration are 2P, 2F, and 4P. Rank these in terms of increasing energy.arrow_forwardFor each of the statements below, indicate whether it is true or false and explain your reasoning. (1 sentence ) 1/ It takes more energy to ionize an electron from the 2s orbital than an electron from the 2p orbital in the Li2+ ion. 2/ The electron affinity of the Ne atom is larger than the electron affinity of the F atom. 3/ K* has a larger radius than Ar. 4/ For a diatomic molecule, in which the internuclear axis is the z-axis, the 3dz? orbital cannot mix with the 2px orbital. 5/ The internuclear distance of O2 increases as it takes an additional electron to become O2".arrow_forwardChemistry This is a molecule of aspirin, a very commonly used painkiller. Draw up a table to show the KEY peaks that you would expect to see in the infrared spectrum of aspirin, identifying which bond has given rise to which peak.arrow_forward
- The vibrations of a CO2 molecule are symmetrical stretch, bend, and asymmetrical stretch (Figure ), with frequencies of 4.02x1013 s-1, 2.00x1013 s-1, and 7.05x1013 s-1, respectively. (a) What wavelengths correspond to these vibrations? (b) Calculate the energy (in J) of each vibration. Which uses the least energy?arrow_forward(a) The ionization energy of molecular hydrogen (H2) is greater than that of atomic hydrogen (H), but that of molecular oxygen (O2) is lower than that of atomic oxygen (O). Explain. (Hint: Think about the stability of the molecular ion that forms in relation to bonding and antibonding electrons.) (b) What prediction would you make for the relative ion- ization energies of atomic and molecular fluorine (F and F2)?arrow_forwardSuppose a diatomic molecule absorbs 200nm light, which excites it into its first excited state. The molecule then dissociates, and 90% of the light energy is converted to kinetic energy. What is the kinetic energy of each atom? What is the total kinetic energy of a mole of these dissociations? Discuss.arrow_forward
- TOPIC: QUANTUM, ATOMIC AND MOLECULAR PHYSICIS a) What are the electron configurations of the molecules 07,02,07 & 0ž- ? b) Determine the symbols for the electronic terms 25+'Au of the ground states of these molecules / g,u ions. Justify your choice of ground state in each case; c) For the case of 02, how do its electronic configuration and term change when a transition occurs from its ground state to its first excited state?arrow_forwardby which formula the exchange energy of the configuration of an atom can be calculated?arrow_forwardTrue or false? The 4d orbital does not exist in the carbon atom. Justify your answer in 1 sentence or 2.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,
Linear Combination of Atomic Orbitals LCAO; Author: Edmerls;https://www.youtube.com/watch?v=nq1zwrAIr4c;License: Standard YouTube License, CC-BY
Quantum Molecular Orbital Theory (PChem Lecture: LCAO and gerade ungerade orbitals); Author: Prof Melko;https://www.youtube.com/watch?v=l59CGEstSGU;License: Standard YouTube License, CC-BY