Write the balanced NET ionic equation for the reaction when Al(NO3)3 and Na3PO4 are mixed in aqueous solution. If no reaction occurs, simply write only NR. Be sure to include the proper phases for all species within the reaction.
Write the balanced NET ionic equation for the reaction when Al(NO3)3 and Na3PO4 are mixed in aqueous solution. If no reaction occurs, simply write only NR. Be sure to include the proper phases for all species within the reaction.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter4: Reactions In Aqueous Solution
Section: Chapter Questions
Problem 78QAP: When 85.0 mL of 0.250 M Ba(OH)2 solution is added to 85.00 mL of 0.250 M Al (NO3)3 solution, a white...
Related questions
Question

Transcribed Image Text:K
o Mail - Thomps
2
8 https://app.101edu.co
P Parchment Rel
3
80
F3
1
Write the balanced NET ionic equation for the reaction when Al(NO3)3 and
Na3PO4 are mixed in aqueous solution. If no reaction occurs, simply write
only NR. Be sure to include the proper phases for all species within the
reaction.
+
$
4
F4
04 03-
2
Reset
TW6D2PHI.pdf
0₂
P
15
%
5
3 4
3
213202
F5
Unofficial Transcri
Question 14 of 40
4
->
Al
O
< 6
A
5
6
7
5
=
0+
F6
UAFS Registra Graduation Applica
6
MacBook Air
²+
&
7
07
NR
N
0
3+
7 8 9 0
09 口。
F7
(s) (1) (g) (aq)
8
4+
•x H₂O
8
CMy.UAFS - Stu
Na
DII
FB
Delete
9
F9
M Inbox (
☆
F1
Expert Solution
Step 1
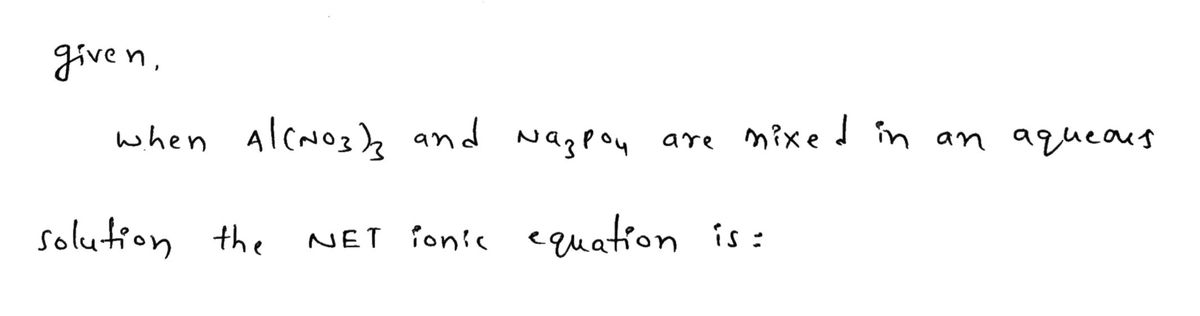
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning