Q: Draw the structure of the aromatic organic product formed from the reaction of the compound with…
A: When an alkyl-substituted benzene is treated with KMnO4, the alkyl part is oxidized to carboxylic…
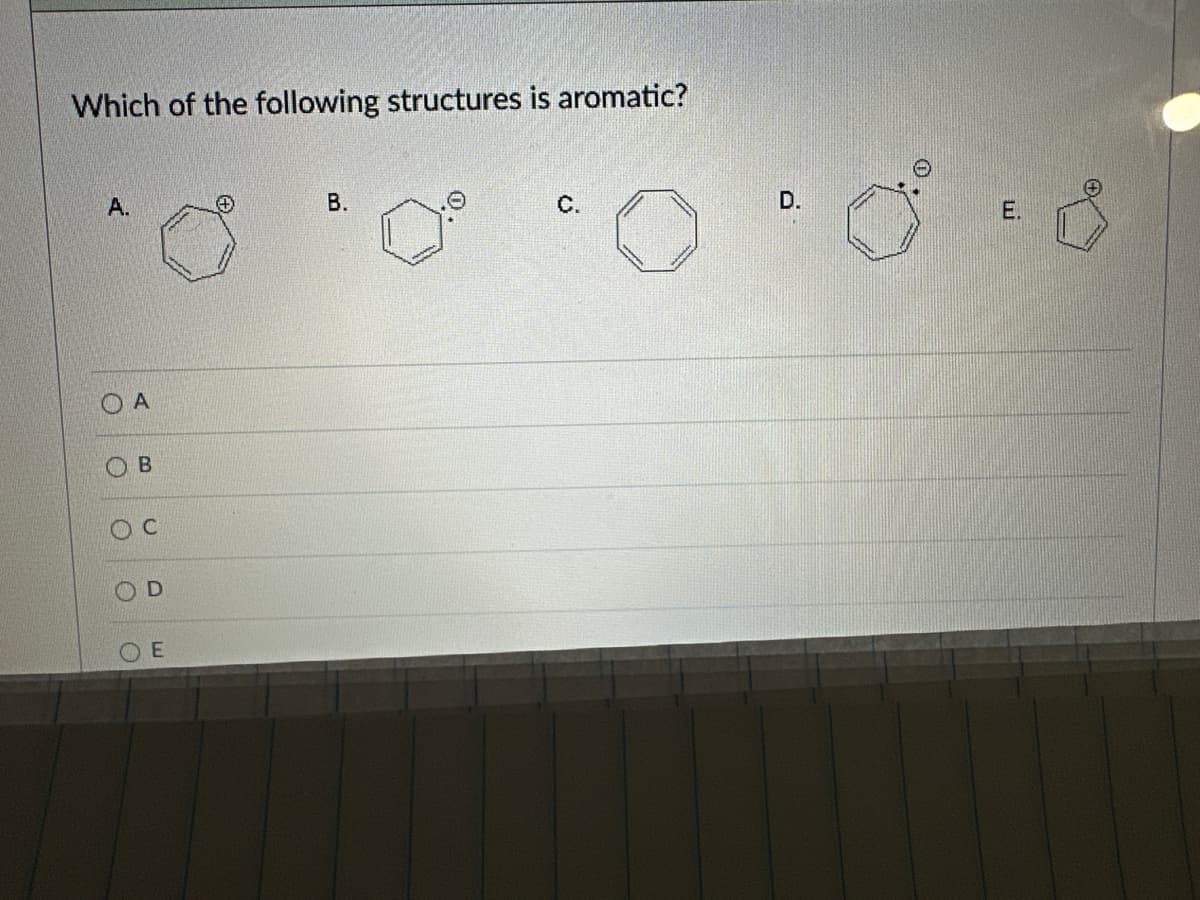
Q: For each of the following structures, would you predict these molecules to possess aromaticity? Why…
A: A planar, cyclic and conjugated molecule is considered to be aromatic if it contains,
Q: Fill in the left side of this equilibrium constant equation for the reaction of hydrofluoric acid…
A: [HF][H3O+][F−]=Ka Explanation:
Q: I need help to draw the particular stage of the mechanism showing this reaction
A: Given reaction is benzil-benzilic acid rearrangement.In this reaction benzil is treated with…
Q: Br 袋 NO2 Pd, PPh3, base NH2
A:
Q: Draw the Molecular Orbital Energy Level Diagram for the following complexes [Cr (CN)6] 4- b. [Cr…
A: The Molecular Orbital Energy Level Diagram represents the molecular orbitals (MO) energy levels in…
Q: Please help me with this homework i tried solving it and it is due today
A: Vant Hoff factor for CaCl2 is 2.43.Vapour pressure of ethanol is 31.7mmHg Vapour pressure of…
Q: from the elements AL, P, MG, Na, Ne, F, O, K ionic compound? draw a lewis dot diagram?
A: Ionic compound formed from the combination of metal and non-metal.Metal atom loss the electrons to…
Q: PyMol commands needed to make the following image of a helix (residues 84-104) found in the Ras…
A: The objective of the question is to generate a specific image of a helix (residues 84-104) found in…
Q: The following compounds, represented with ball-and-stick models, have the same molecular formulas…
A:
Q: Please don't provide handwritten solution ...
A: the major product will be the conjugated diene. Explanation:We know that in the presence of the base…
Q: please help!!!! I need a detailed reaction mechansim for this molecule and the name of the molecule!…
A: Esterification is a chemical reaction in which two reactants, typically an alcohol and an acid,…
Q: Determine the pH during a titration of 25.0 mL of 0.5 M HI with 25.0 mL of 0.5 M KOH.
A: Given,molarity of HI = 0.5 MVolume of HI = 25.0 mLMolarity of KOH = 0.5 MVolume of KOH = 25.0 mL
Q: Write the precipitation reaction for calcium bromide in aqueous solution: (Use the lowest possible…
A: We have the precipitation reaction forcalcium bromide, we are to write the balanced equation for…
Q: Students were asked to find the identity of a wooden object. The mass of the object is 33.0 grams. A…
A: given that = dimension of object is 3.0cm*5.2cm*2.8cmmass of the object = 33.0 gramsdensity is a…
Q: H3C What is the product of the lithium aluminum deuteride (an isotopic alternative to hydrogen)…
A: This is an example of reduction of ester to primary alcohol
Q: Consider the reaction below. Which species is (are) the Brønsted-Lowry acid(s)? HF (aq) + NH (aq) =…
A: A. HF, NH4+Explanation:Approach to solving the question:Bronsted-Lowry is a theory for certain…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. CH3CH2CH2NH2, Na(CN)BH3, pH…
A: Reductive amination involves the reaction of carbonyl compound with amine to form an imine which is…
Q: ← Problem 22 of 30 Submit Draw the major product of this reaction. Ignore inorganic byproducts. H 1.…
A: Tollens’ reagent is a colorless, basic, aqueous solution containing silver ions coordinated to…
Q: Suggest reaction conditions or short synthetic sequences that could provide the reactant from…
A: The objective of the question is to suggest reaction conditions or short synthetic sequences that…
Q: Calculate the solubility at 25 °C of AgBr in pure water and in 0.19 M NaI. You'll probably find some…
A: Temperature = Molarity of NaI = 0.19 M
Q: By using only amide/Peptide bond forming rxn and a lKylation rxns. Provide all the reaction steps…
A: The objective of the question is to outline the steps for forming a product using only amide/peptide…
Q: reasonable reaction sequence to use to synthesize it.
A: Given is organic synthesis reaction. The given starting compounds have functional groups like…
Q: Add the missing nucleophile to the substitution reaction in the drawing area below. Note for…
A: Answer:In the given reaction one nucleophile is being substituted by the other nucleophile and…
Q: Using the table of indicators identify which of the given indicators would be appropriate for the…
A: Answer:Indicator is the chemical substance that indicates the completion of titration by changing…
Q: H H H3CO Li Na2Cr2O7 H2SO4
A: Given reaction:We have to find the missing products
Q: e major product of this condensation reaction. Ignore inorganic byproducts. NO₂ 1. NaOH, heat 2.…
A: The objective of the question is to find the major product of the given condensation reaction in…
Q: Use the balanced equation for the reaction of F2 and N2 to form NF3 How many moles of NF3 are formed…
A: The objective of the question is to determine the number of moles of NF3 that can be formed from 1.0…
Q: 22. Draw the major product of the following SN2 reaction. Draw a mechanism, showing the transition…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: has the correct number of significant digits. 12 22 11, Note: Reference the Vapor pressure of water…
A: (Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: 2) Draw the major product expected for the reaction below: Me2NH (excess) acetic acid
A: The objective of the question is to find the expected major product of the reaction in which…
Q: Part IV: 1. Provide the mechanism and product for the reaction shown below. ONa Br DMF 2. Provide…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: Draw the mechanism and 1) consider the two halves of the naphthalene as separate - which is more…
A: The objective of the question is to understand the reactivity of the two halves of naphthalene and…
Q: Draw structural formulas for the a.ß-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: It is an organometallic reagentcompound having general formula R2CuLi. R groups are alkyl, aryl or…
Q: In each case below select the synthetic procedure/s that could be used to carry out the…
A: Hydroboration and oxymercuration are two distinct processes used in chemical synthesis to add…
Q: The Ksp for the insoluble ionic compound with formula of M(OH)3, in which M represents a metal is:…
A: Since,Solubility product constant is the multiplication of the dissociated ion with the power of…
Q: What would be the product of the following reaction sequence? 0 1. DIBAL-H, -78°C. 2. H₂O OH ? & are…
A: Given,The reaction is:
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate the…
A: The given reaction is between an alkene and catalytic OsO4 in presence of NMO. OsO4 when reacts…
Q: "Synthesis gas" is a mixture of carbon monoxide and water vapor. At high temperature synthesis gas…
A: Given,
Q: 5) Suggest a series of reagents that could be used to carry out the following transformation. More…
A: Grignard reagent is an organometallic reagent which involves C-Mg bonding. It is represented as…
Q: A. 2 H NaOH
A:
Q: Put the following leaving groups (LG) in decreasing order of their ability to be eliminated from the…
A:
Q: E 1. CH,MgBr 2.HIO* 1. Na(CN)BH, pH 4-5 2. HO CHÍNH, NGOH 1. CHICH₂NH2, Na(CN)BHz. pH 4-5 2.…
A: Find out suitable reagent for conversion of the reaction Alkyl cyanide into ketone
Q: OH 1. NaOH, EtOH 2. n-BuBr A nucleophilic substitution reaction is a frequently used method to…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Content/24SSEM_CHEM_114 X Aktiv Chemistry - Work b Home | bartleby x + ← → с app.aktiv.com Question…
A: The objective of the question is to determine which electron transition in a hydrogen atom would…
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A: Since the central atom has four bonded atoms or groups of atoms attached to it, it confirms the…
Q: Draw a plausible mechanism for the following transformation: [H2SO4] excess EtOH -H₂O EtO OEt
A: When ketone is treated with excess of alcohol in presence of acid catalyst forms a carbon with two…
Q: Draw the structure of the organic reactant, and write the chemical formula of the reagent used to…
A: Given is organic reaction.The given product is 2,6-dinitroanisole.So, starting compound of this…
Q: Calculate the [H3O+] of a lime juice solution that contains [OH-] = 9.10\ times 10-12 M. Show work.…
A: We need to calculate concentration of hydroxide ions. We have been provided with hydronium ion…
Q: 2) Suppose a student obtains a 56.55 mL sample of an unknown (monoprotic) weak acid, and proceeds to…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Step by step
Solved in 1 steps

- The purine heterocycle occurs commonly in the structure of DNA.a.How is each N atom hybridized? b.In what type of orbital does each lone pair on a N atom reside? c.How many π electrons does purine contain? d.Why is purine aromatic?Which of the following compounds is aromatic? A. only A and B B. only A and C C. only B and C D.only B1. (a) Describe aromaticity, Kekule structure and resonance structure for benzene. (b) Why is benzene more stable than aliphatic alkenes?
- 1. Thiophene,furan and pyrrole which is more aromatic and reactive? 2. Heterocyclicgroups in haemoglobin,folic acid, morphine and caffeine.Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with HNO3, H2SO4.Tell whether the final products in the given reactions are aromatic or not?
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with isobutylene and HFExplain the relevance pi* (antibonding) -molecular orbitals have in nucleophilic addition reactions of carbonyl containing compounds.Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hckel criteria? Which, if planar, would be antiaromatic? 21.15 State the number of 2p orbital electrons in each molecule or ion.
- N-Phenylsydnone, so-named because it was first studied at the University of Sydney, Australia, behaves like a typical aromatic molecule. Explain, using the HĂ¼ckel 4n + 2 rule.Compound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?Which (if any) lone pairs are participating in aromaticity?









