At 2000 °C the equilibrium constant for the reaction 2NO(g) = N₂(g) + O2(g) is Ke 2.4 x 10³. ▼ Part A If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of NO? Express your answer to two significant figures and include the appropriate units. -4.8.10² Submit Part B O Value 1 X Incorrect; Try Again; 4 attempts remaining Submit Part C M If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of N₂? Express your answer to two significant figures and include the appropriate units. A Previous Answers Request Answer wwwww ? Units Request Answer 國」? If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of O₂? Express your answer to two significant figures and include the appropriate units.
At 2000 °C the equilibrium constant for the reaction 2NO(g) = N₂(g) + O2(g) is Ke 2.4 x 10³. ▼ Part A If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of NO? Express your answer to two significant figures and include the appropriate units. -4.8.10² Submit Part B O Value 1 X Incorrect; Try Again; 4 attempts remaining Submit Part C M If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of N₂? Express your answer to two significant figures and include the appropriate units. A Previous Answers Request Answer wwwww ? Units Request Answer 國」? If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of O₂? Express your answer to two significant figures and include the appropriate units.
Chapter13: Chemical Equilibrium
Section: Chapter Questions
Problem 108CP: Consider the decomposition equilibrium for dinitrogen pentoxide: 2N2O5(g)4NO2(g)+O2(g) At a certain...
Related questions
Question

Transcribed Image Text:At 2000 °C the equilibrium constant for the reaction 2NO(g) = N2(g) + O2(g) is
Kc = 2.4 x 10³.
Part A
If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of NO?
Express your answer to two significant figures and include the appropriate units.
-4.8.10²
Submit
Part B
X Incorrect; Try Again; 4 attempts remaining
Value
Submit
If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of N₂?
Express your answer to two significant figures and include the appropriate units.
Part C
0
UA
M
Previous Answers Request Answer
Submit
Value
ΠΑ
Units
Request Answer
If the initial concentration of NO is 0.175 M, what is the equilibrium concentration of O2?
Express your answer to two significant figures and include the appropriate units.
www ?
Units
Request Answer
?
****
?
Expert Solution
Step 1
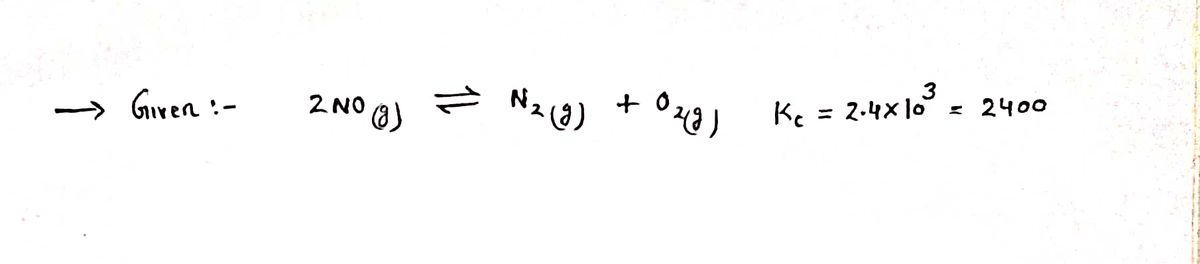
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning