A chemistry graduate student is studying the rate of this reaction: 2SO3(g) 2SO₂(g) + O₂(g) She fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds: time (seconds) [s03] 0 0.0100M 10. 0.00385M 20. 0.00238M 30. 0.00172M 40. 0.00135M Use this data to answer the following questions. Write the rate law for this reaction. Calculate the value of the rate constant k Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol. rate = k k = 0 x10 0.0 X 2 3
A chemistry graduate student is studying the rate of this reaction: 2SO3(g) 2SO₂(g) + O₂(g) She fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds: time (seconds) [s03] 0 0.0100M 10. 0.00385M 20. 0.00238M 30. 0.00172M 40. 0.00135M Use this data to answer the following questions. Write the rate law for this reaction. Calculate the value of the rate constant k Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol. rate = k k = 0 x10 0.0 X 2 3
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 100AE: Consider a hypothetical reaction between A and B: A + B products Use the following initial rate...
Related questions
Question
100%
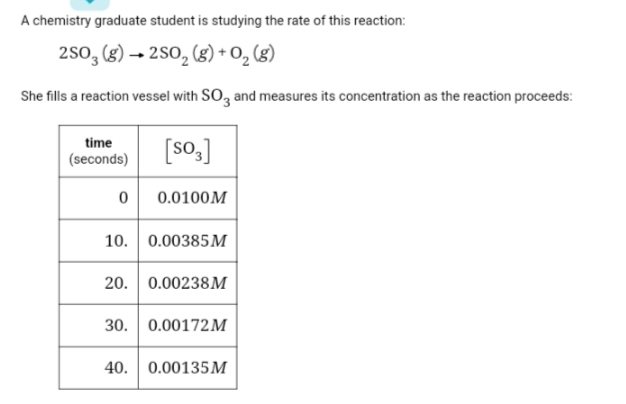
![A chemistry graduate student is studying the rate of this reaction:
2SO3 (g) → 2SO₂(g) + 0₂ (8)
She fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds:
time
(seconds)
[SO3]
0 0.0100M
10. 0.00385M
20. 0.00238M
30. 0.00172M
40. 0.00135M
Use this data to answer the following questions.
Write the rate law for this reaction.
Calculate the value of the rate constant k
Round your answer to 2 significant digits. Also be sure
your answer has the correct unit symbol.
rate = k
k = 0
x10
ロ・ロ
X
2
200
Ar](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F787afdef-39b0-48af-8625-675705fdb4a7%2Fbb352479-8a07-4494-8584-ddd4f517f240%2Ffcijkh_processed.jpeg&w=3840&q=75)
Transcribed Image Text:A chemistry graduate student is studying the rate of this reaction:
2SO3 (g) → 2SO₂(g) + 0₂ (8)
She fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds:
time
(seconds)
[SO3]
0 0.0100M
10. 0.00385M
20. 0.00238M
30. 0.00172M
40. 0.00135M
Use this data to answer the following questions.
Write the rate law for this reaction.
Calculate the value of the rate constant k
Round your answer to 2 significant digits. Also be sure
your answer has the correct unit symbol.
rate = k
k = 0
x10
ロ・ロ
X
2
200
Ar
Expert Solution
Step 1
Given that,
We have to find out the rate equation and the rate constant value.
Introduction: Rate law.
Formula: The integrated rate law for the second-order reaction is .
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning