3) 2.413 g sample contains Na CO,, NaHCO, and inert material. This sample was solved in water and diluted to 250.0 ml. 80 ml solution was taken and titrated with 0.09644 N HCl with the indicator of phenolftalein. 6.13 ml HCl was consumed. From a second solution, again 80 ml was taken and titrated with the same HCl solution with the indicator of methylorange. 15.4 ml HCl was consumed. Calculate the percentage of NaHCO, and Na CO in sample (Na CO=106.0 g/mol, NaHCO =84.02 g/mol).
3) 2.413 g sample contains Na CO,, NaHCO, and inert material. This sample was solved in water and diluted to 250.0 ml. 80 ml solution was taken and titrated with 0.09644 N HCl with the indicator of phenolftalein. 6.13 ml HCl was consumed. From a second solution, again 80 ml was taken and titrated with the same HCl solution with the indicator of methylorange. 15.4 ml HCl was consumed. Calculate the percentage of NaHCO, and Na CO in sample (Na CO=106.0 g/mol, NaHCO =84.02 g/mol).
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 26P
Related questions
Question
100%

Transcribed Image Text:3) 2.413 g sample contains Na CO,, NaHCO, and inert material. This sample was solved
in water and diluted to 250.0 ml. 80 ml solution was taken and titrated with 0.09644 N
HCl with the indicator of phenolftalein. 6.13 ml HCl was consumed. From a second
solution, again 80 ml was taken and titrated with the same HCl solution with the indicator
of methylorange. 15.4 ml HCl was consumed. Calculate the percentage of NaHCO, and
Na CO in sample (Na CO=106.0 g/mol, NaHCO =84.02 g/mol).
Expert Solution
Step 1
Following are the two reactions and their indicators can be represented as:
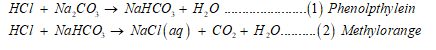
Sample contain
Na2CO3 + NaHCO3 + Inert Material = 2.413 g in 250 mL water.
Reaction 1 for Na2CO3:
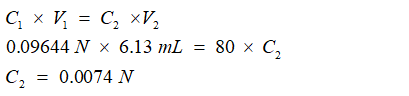
Step 2
Now calculate the number of moles of sodium carbonate in 250 mL sample solution:
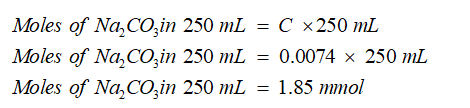
Now calculate weight of sodium carbonate in the sample as:
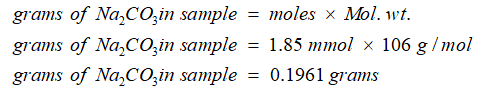
Step 3
Now percentage of Na2CO3 can be calculated as:
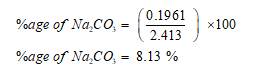
Reaction 2 for combine Na2CO3 and NaHCO3
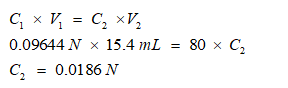
Note: first Na2CO3 convert into NaHCO3, therefore next there will be 3 times of the NaHCO3, two from Na2CO3 and 1 from NaHCO3
Step by step
Solved in 6 steps with 9 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning