Q: When 93.0 g of calcium and 45.5 g of nitrogen gas undergo a reaction that has a 91.1% yield, what…
A:
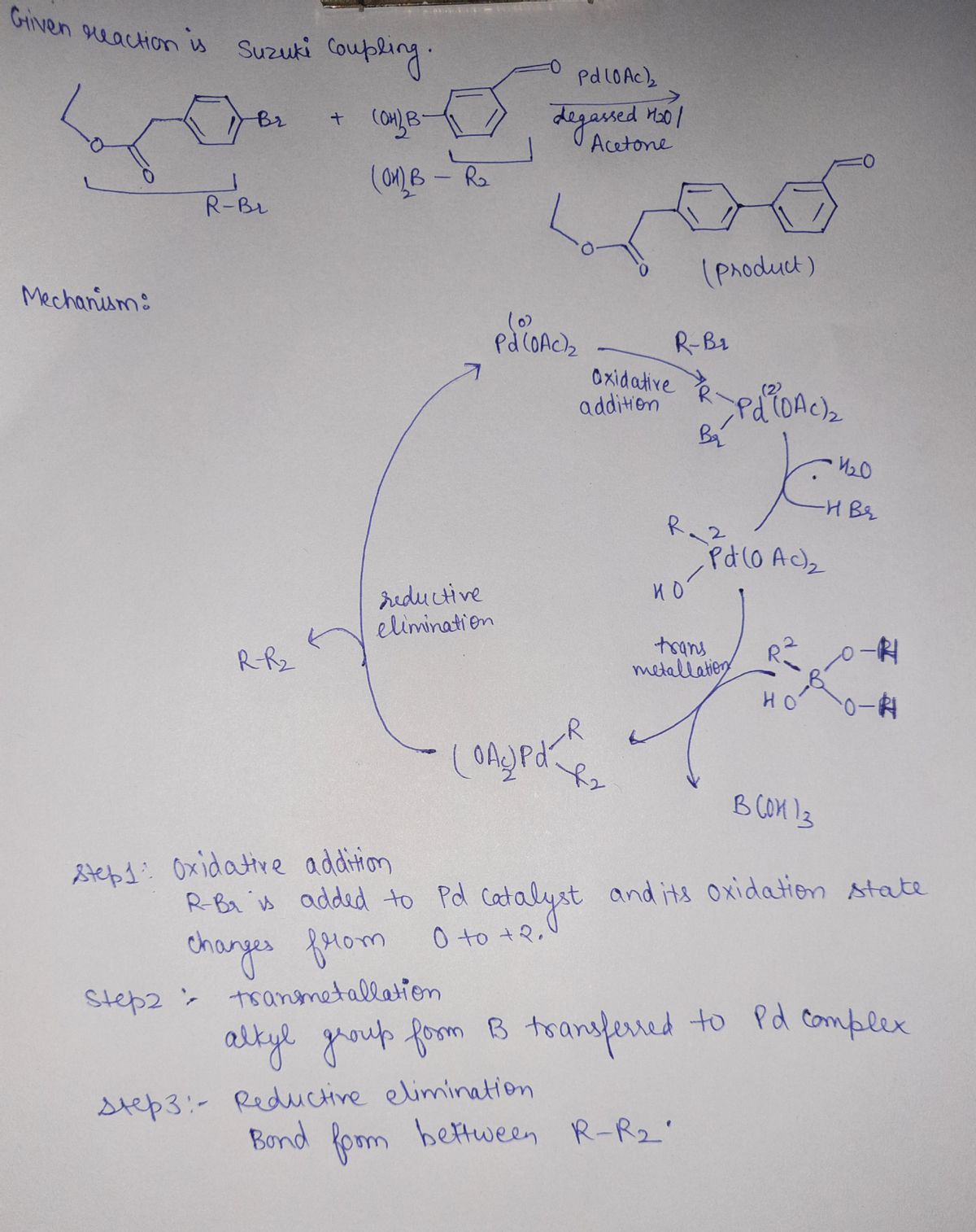
Q: 2 3 5 6 Predicate the products of each reaction: Aldehydes + Ketones CH;CH + HC=N o O CH₂-C-OCH, =O…
A: Here, we have to draw the products for the given organic reactions.
Q: What is the energy of attraction (in joules) between a cation with a valence of +2 and an anion with…
A: “Since you have asked multiple question, i will solve the first question for you. If you want any…
Q: 12. Consider the reaction 4PH3(g) →→→ P4(g) + 6H₂(g) - If, in a certain experiment, over a specific…
A: Given -> 4PH3(g) ---> P4(g) + 6H2(g) Moles of PH3 consumed = 0.0048 mole Volume= 2.0 L
Q: Phosphorus and bromine react to form phosphorus tribromide, like this: P4(g)+6 Br₂(g) →4 PBr3(9)…
A: The given equilibrium reaction is as follows: P4(g) +6Br2(g) ⇌ 4PBr3(g) The pressure equilibrium…
Q: Problem 1. Propose a reasonable synthesis for the following compound from methylcyclohexane. (Hint:…
A: In this question, we will synthesized the final product from the given starting material. We will…
Q: a) SO₂ gas at a pressure of 7.50 atm is heated from 0.0°C to 481 °C and is simultaneously compressed…
A:
Q: Macmillan Learning If the K, of a monoprotic weak acid is 3.3 x 10-6, what is the pH of a 0.41 M…
A:
Q: What is the activity coefficient for each ion at the given ionic strength at 25 °C? Activity…
A: Activity Coefficients: Definition: To account for the deviation from ideal behavior due to the ionic…
Q: Why is Cl₂ + KI - KCI + 12 not a synthesis reaction?
A: Given : about synthesis reaction Tip : Focus on the definition of synthesis reaction
Q: When a small amount of water is added to t-butyl, what happens to the cooling curve for this new…
A: Colligative properties are properties of a solution that depend on the concentration of solute…
Q: This graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure,…
A: We have find out the answer.
Q: The effect of intermolecular forces on a gas becomes more pronounced at relatively greater deviation…
A:
Q: Four liquids are described in the table below. Use the second column of the table to explain the…
A: Given 2.1 g potassium nitrate in 150. mL water 2.1 g of potassium chloride in 150 mL water 2.1 g HI…
Q: Determine the specific heat of a 2.58 g/mL block at 100 °C that causes the level of 0 °C water to…
A: The specific heat of the block is given by the formula, Specific heat =Heat released by the…
Q: The proton concentrations of three solutions at 25 °C are given. Classify the solutions as acidic,…
A: According to the proton concentrations given, we have to determine if the solution is acidic, basic…
Q: A volume of 500.0 mL of 0.130 M NaOH is added to 605 mL of 0.200 M weak acid (Ka = 3.87 × 10¯). What…
A: Given -> Volume of NaOH= V1 = 500 ml Molarity of NaOH = M1 = 0.130 M Volume of weak acid = V2 =…
Q: An automobile engine generates 2210 Joules of heat that must be carried away by the cooling system.…
A: Please find your solution below : According to the first law of thermodynamics, energy can neither…
Q: What is the by-product of the following reaction? NH₂ 8. + a. H₂O b. C.Hi C. H 10 i d. HO ii Select…
A: Identify the product and by-product of the following incomplete reaction.
Q: O + 2 A
A: When a conjugate diene reacts with dienophile in presence of heat they undergo cyclo addition…
Q: Consider a solution is prepared by dissolving 34.0 g of NH3 in 500.0 g of water. The density of the…
A:
Q: How many milligrams of solid MgCl2 (Formula Weight: 95.211 g/mole) are required to prepare 250mL of…
A: Given : formula weight = 95.211 g/mol Volume = 250 mL Molarity = 150 mM
Q: Enter your answer in the provided box. What is the wavelength (in meters) of an electromagnetic wave…
A: Given: Frequency (ν) of electromagnetic wave = 1.27 x 1012 s-1 To determine the wavelength (in…
Q: What will be the product or products if zinc (Zn) reacts with hydrochloric acid (HCI) Most active…
A:
Q: The following table contains some data showing the effect of molar concentration of a solution on…
A: Given: The data for concentration and optical density is: Concentration (M) Optical density…
Q: Please draw 2-propylpentane and 4-propylpentane.
A: Please find your solution below : Structural formula is the representation of a compound in which…
Q: 4. Select all that is considered spontaneous a. Cleaning your room b. A piece of bread molding after…
A: A spontaneous process is a process that occurs without the need for an external influence and…
Q: Which drawing best represents the resonance hybrid for the given molecule? H₂C-C=N 8- H₂C----C=N 8 8…
A: In the drawing of the resonance hybrid, bond breaking and bond forming denoted by the doted line.
Q: Draw the two products of the reaction shown below. Ignore inorganic byproducts.
A: Given : structure of reactant
Q: What is the correct IUPAC name of the following compound? H H CH₂CH3 CH₂ Multiple Choice…
A: Introduction IUPAC nomenclature is a system of naming chemical compounds and identifying the…
Q: 2) For the reaction 2A + B →3D, which statement(s) below is/are true? Circle all that apply. a. The…
A: This question is related to chemical kinetics. Chemical kinetics is a branch of chemistry that…
Q: 9) Which of the following compounds does not have the molecular formula C6H₁4O? (a) 1-hexanol (b)…
A: The molecular formula is the expression of the number of atoms of each element in one molecule of a…
Q: What kind of intermolecular forces act between a hydrogen iodide molecule and a chloroacetylene…
A: Introduction Intermolecular forces are the forces of attraction or repulsion that act between…
Q: A. PHYSICAL PROPERTIES OF ELEMENTS AND COMPOUNDS Using the information obtained during the…
A: Element is pure form of substance consist only one type of atoms
Q: Fill in the orbital energy diagram for nickel. 4s E 3s- 2s 1s 3p- 2p- 3d AV AV The lowest E levels…
A: This question is related to atomic structure. In the given question, it is asked to draw orbital…
Q: For each solute, click the button under the better solvent. solute :0: || NH,−C. NH Br NH₂ HO · CH₂…
A: Solubility of a substance in a solvent follows "Like dissolves like " which means if any solvent is…
Q: What would be the approximate H-C-H bond angle in CH3OH? (In other words, what is the approximate…
A:
Q: The reaction A→ B has a standard state free energy of -15 kJ/mol and a cellular free energy of 0…
A: Introduction Spontaneous reaction in chemistry is a process in which a chemical reaction proceeds…
Q: Decide for which of the following compounds cis-trans isomerism is possible and draw the trans…
A: Given : structure of molecules
Q: Where did you get the 1.6*10^-19 and the 9.1*10^-31?
A: This question is related to atoms. There are mainly three subparticles in an atom- Electron Proton…
Q: Iron-59 has a half-life of 45 days. If 168 g of a radioactive iron (59Fe) is received in the lab…
A:
Q: What concentration of barium hydroxide is needed to give an aqueous solution with a pH of 12.430?…
A: Answer:- This question is answered by using the simple concept of calculation of molarity using the…
Q: There are two four-carbon branched-chain alkane constitutional isomers True or false
A: Answer: Two compounds having same molecular formula but different structural formulas are called as…
Q: 17.3 Suggest reagents for the transformation. shown below. HO
A: -> In Markovnikov addition nucleophilic part attached to that double bonded carbon which has…
Q: Which best describes the resonance hybrid for the following molecule? N The electron deficiency is…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: Can you help me with the explanation of the noncovalent interaction, hydrogen bonding, dipole-dipole…
A:
Q: Which chemical reaction below yields the heat of formation of liquid ammonia (NH3)? 3H(g) + N(g)→→…
A: Reaction equation in which a mole of a chemical compound is formed from its constituent elements…
Q: Calculate the molarity of a 1.03 m aqueous solution of tin(IV) nitrate with a density p= 1.082 g/mL.…
A:
Q: Indicate whether each of the following compounds will give an acidic, basic or neutral solution when…
A: Answer: This question is based on hydrolysis of salt solutions which help us to decide whether a…
Q: At a particular temperature, N₂O5 decomposes according to a first-order rate law with a half-life of…
A:

Step by step
Solved in 2 steps with 1 images

- Which statement best describes the products of this reaction?potassium dichromate (Cr2O7^2-) oxidation of ethanol to acetic acid is the basis for the original breath-alcohol screening test used by law enforcement agencies to determine a person's blood alcohol content (BAC). the test is based on the difference in colour between the dichromate ion( redish orange) in the reagent and the chromium(iii) ion (green) in the product. deduce the reaction that is involved in this test and give the IUPAC name of the other product formed.What is the major product in the following reaction? Br ·0-
- Complete the following equations. If no reaction occurs, write no reaction. a. b. c. d. e.What would be the product(s) of following reaction? 3 Feo + 2 AIC13 → ? 3FECI, + Al203 FeCl3 + AlO O FezAlhO3Cl2 O FezAl, + 30C12We have a hydrocarbon, C16H26. The compound has two triple bonds and the acids produced by ozonolysis are CH3(CH2)4CO2H and H2OCCH2CO2H. What is a likely structure for this hydrocarbon?

