
Concept explainers
Which block in each of the following drawings of a balance is more dense, red or green? Explain.
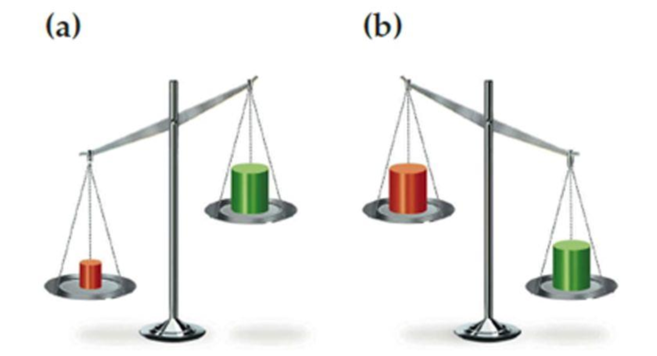
a)
Interpretation:
The denser block in drawing of a balance has to be identified and explained.
Concept Introduction:
Density:
Density can be defined as property that links mass of an object to its volume. It derived quantity and its unit is
Explanation of Solution
It is known that,
Density is directly proportional to mass and inversely proportional to volume.
In (a), red is identified as denser block because it is found have more mass and less volume than green block.
b)
Interpretation:
The denser block in drawing of a balance has to be identified and explained.
Concept Introduction:
Density:
Density can be defined as property that links mass of an object to its volume. It derived quantity and its unit is
Explanation of Solution
It is known that,
Density is directly proportional to mass and inversely proportional to volume.
In (b), green is identified as denser block because it is found have more mass and less volume than green block.
Want to see more full solutions like this?
Chapter 0 Solutions
General Chemistry: Atoms First
- 1.82 Which of the following molecular-scale diagrams best represents a pure compound? Explain your answer.arrow_forwardSeawater is composed of salt, sand, and water. Is seawatera heterogeneous or homogeneous mixture? Explain.arrow_forwardYou receive a mixture of table salt and sand and have to separate the mixture into pure substances. Explain how you would carry out this task. Is your method based on physical or chemical properties? Explain.arrow_forward
- Calculating the density of a rectangular wooden block (fig 3). In determining the density of a rectangular wooden block, use the information in the table below and the formula to fill in the empty boxes. (Apply the uncertainty rule) Fig 3. Volume of wooden block (cm) = Length (cm) x Width (cm) x Thickness (cm) Mass (g) of wooden block (g) Length of wooden block (cm) Width of wooden block (cm) Thickness of wooden block (cm) Volume of wooden block (cm) Density of wooden block (g/cm) d= m/v 20.5g 5.00cm 5.00cm 1.00 cm 2arrow_forwardDirection: Identify each mixture as either homogeneous or heterogeneous. 1. Orange juice 2. Smoke 3. Paint 4. Soda 5. Steelarrow_forwardClassify these substances. More than one answer may apply in each case. H2 pure substance element element homogeneous mixture heterogeneous mixture compound homogeneous mixture solution solution heterogeneous mixture compound O pure substance H,0 salt water heterogeneous mixture homogeneous mixture O pure substance Oheterogeneous mixture O solution U pure substance homogeneous mixture I element compound O solution element O compound 口□ 口口ㄩarrow_forward
- classify whether it is greater than, less than, or equalarrow_forwardHow can you distinguish an element, from a compound, from a solution, from a heterogeneous mixture? Explainarrow_forwardHow many grams F in 650.1 g Br2F6? Put the answer in the box in regular (non-scientific) notation. Use one figure past the decimal.arrow_forward
- Liquid 1 reacts with Liquid 2, producing a solid and a gas. Using this scenario, which supports the law of conservation of mass? mass of Liquid 1+ mass of solid = mass of Liquid 2 + mass of gas mass of Liquid 1- mass of solid = mass of Liquid 2 – mass of gas mass of Liquid 1- mass of Liquid 2 = mass of solid + mass of gas mass of Ligquid 1+ mass of Liquid 2 = mass of solid + mass of gas Next Submit Save and Exit Mark this and return Sign ouarrow_forwardThe mass of magnesium used in this experiment is critical to determining the moles of hydrogen gas generated. The analytical balances used in Chemistry 1A labs are very sensitive to point that touching materials with your fingers before measuring them can affect the measurements. The balances are sensitive enough to detect dirt and oils from your fingers transferred to the magnesium. Would your experimental value of R increase or decrease if the mass of magnesium measured was artificially high due the touching it with your hands? Increase Decreasearrow_forwardWater has a density of 1.00g/mL. How many teaspoons of water are represented by 40,500 molecules of water. Use dimensional analysis.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



